Non-Clastic Rocks and Their Classification
Non-Clastic Rocks and Their Classification
Carbonate Rocks
- Carbonates are the most abundant non-terrigenous sedimentary rocks.
- In contrast to terrigenous rocks, they form chemically and biochemically. Dissolved ions carried from source to depositional site in solution eventually precipitate and form solid minerals.
- The distinction between biochemical and chemical sedimentary rocks is not always clear-cut.
- Organisms have a role in the formation of many sedimentary rocks. For example, they can extract dissolved components from seawater to manufacture shells or skeletons that later are incorporated into sedimentary rocks.
- Organisms can also be indirectly involved, such as when their metabolism modifies the geochemical setting enough to cause mineral precipitation.
- The distinction between clastic sedimentary rocks and "crystalline" chemical and biochemical rocks such as carbonates can also be ambiguous.
- Many limestones contain composite grains formed of an interlocking mosaic of calcite or aragonite.
- These composite grains include ooids and intra-basinal detrital carbonate called intra-clasts.
The Importance of Limestone
- Limestone tells us much about the origin and evolution of the Earth.
- The texture and composition of limestone reveal as much about depositional setting as do the texture and composition of sandstone, conglomerate, and breccia.
- Limestones are the most important evidence remaining of shallow marine seas that covered much of the world during most of the Phanerozoic.
- They are almost invariably fossiliferous they are probably the best documentation of organic evolution.
- They dissolve easily, they are often porous in the subsurface and become reservoir rocks for petroleum and natural gas carbonate rocks are important aquifers.
- High porosity also makes limestones excellent host rocks for ore-bearing solutions (require an inexpensive base to neutralize acid.
- Limestone deposition in the oceans is partially responsible for Earth's unique atmosphere.
- Without this process and organic evolution, Earth's atmosphere would resemble those of Venus and Mars (mostly carbon dioxide).
- Two contrasting states of the Earth, icehouse and greenhouse, produce dramatic differences in seawater temperature and chemistry.
- During much of the Phanerozoic warm greenhouse conditions prevailed. There were no polar ice caps, sea level was higher, the oceans were less stratified, and enormous volumes of limestone could be deposited.
- Under cold icehouse conditions (the Permo-Triassic, the late Cenozoic, and presently), sea level is low, there are extensive polar ice caps, ocean water is stratified, and little carbonate is formed.
- The mineralogy of carbonates also rejects ocean water conditions, with high-Mg calcite precipitated under greenhouse conditions and aragonite and low-magnesium calcite formed under icehouse conditions.
Carbonate Mineral Chemistry
- All carbonate minerals are formed by combining divalent cations (2+ )-particularly calcium and magnesium, as well as minor amounts of iron, strontium, manganese, and barium-with carbonate anions (CO3)2-
- Three are abundant in the Earth's crust (or at the surface): calcite, aragonite, and dolomite.
- The first two are polymorphs of CaCO3 Calcite is a soft mineral that readily fizzes in acid.
- Aragonite is less stable than calcite under most conditions and tends to alter into calcite during diagenesis.
- Dolomite, CaMg(CO3)2 is the third important carbonate mineral.
- The structure of dolomite consists of alternating layers of Ca and Mg ions separated by layers of C03.
- Ideally, it should have equal amounts of Ca and Mg, Ferrous (Fe2+) iron also substitutes for the magnesium.
- If enough ferrous iron is present, it forms ferroan dolomite or ankerite, with the composition Ca(MgFe)(CO3)2.
Controls on Carbonate Deposition
- Three physical conditions control how much C02 can be dissolved: temperature, pressure (water depth), and degree of agitation.
- Temperature Raising the temperature of seawater promotes limestone deposition form more readily in tropical seas than in polar waters.
- Pressure Reducing the pressure (or depth) of seawater promotes limestone deposition. Modern carbonate sediment and ancient limestone form more readily in shallow water than in deep water.
- Degree of agitation Breaking waves in the surf zone mix seawater with air. This agitation promotes limestone formation because additional carbon dioxide is absorbed by the atmosphere but four other factors are also important: organic activity, sediment masking and clogging, light, and carbonate compensation depth.
- Organic activity Plants and animals either precipitate calcium carbonate directly or modify the geochemical environment enough for precipitation to occur Plants such as phytoplankton and algae promote calcium carbonate precipitation because they remove carbon dioxide from seawater.
- Sediment masking and clogging the kind of sediment accumulating at any point in time and space reflcts what isn't happening as much as what is; this is the sedimentary masking effect.
- Light. Because photosynthetic organisms (especially calcareous algae and hermatypic corals) require light for photosynthesis, most large carbonate accumulations form in water shallow enough (less than 20 m deep) for adequate light to penetrate.
- Carbonate compensation depth (CCD). The temperature and pressure of very deep seawater control the areal distribution of calcareous ooze on the modern abyssal plains.
- Calcareous ooze consists of the unconsolidated shells of floating pelagic organisms that thrive in the photic zone that extends from the water surface to about 200 m.
- After organisms die, their shells settle to the seafloor.
- Several factors control the rate at which calcareous ooze accumulates.
- The rate at which shells are supplied from above is very important and reflects organic productivity.
- This rate varies with water temperature and is largely a function of latitude, water depth, and the supply of organic nutrients.
- Equally important is the rate at which shells are destroyed by dissolution. This is controlled by the carbonate compensation depth.
- The CCD is analogous to the permanent snow line in mountains, which is simply the elevation above which there is year-round snow cover.
- The CCD marks the water depth at which slowly falling calcium carbonate sediment dissolves at precisely the same rate as it is supplied from above.
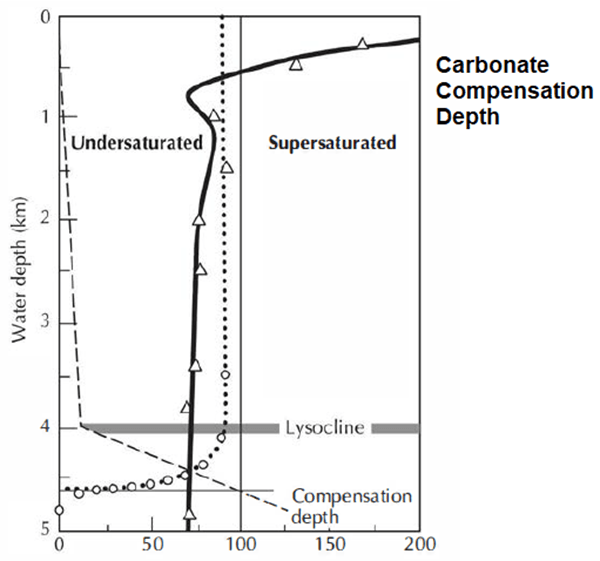
- Calcareous oozes accumulate only above the CCD; below it, calcium carbonate dissolves at a faster rate than it is supplied Colder temperatures increase the rate of solution.
Limestone Components and Classifications
- The most useful schemes for classifying limestone recognize the diverse origin of carbonate rocks and the elastic aspect of their texture.
- One of the most popular classifications was devised by R. L. Folk.
- It separates allochemical and orthochemical components.
- Allochemical components (or allochems) are any grains of calcium carbonate that, after formation, are transported and deposited as clasts. sandstone.
- There are different types of allochems, including coated grains (such as ooids) and skeletal fragments (bioclasts).
- Orthochemical components (or orthochems) are not transportedand deposited as clasts.
- Orthochems are found precisely where they formed or have been moved only a short distance.
- There are two kinds of orthochems: (1) microcrystalline calcite matrix, or micrite, is fine-grained (finer than 4 microns in diameter) carbonate mud, analogous to matrix in wacke sandstone; (2) microcrystalline sparry cement, or spar, is relatively clear interlocking crystals of calcium carbonate, analogous to cement in arenite sandstone.
- Figure shows the four principal limestone families: (1) sparry allochemical rocks, (2) microcrystalline allochemical rocks; (3) micrite; and (4) biolithite.

- Sparry allochemical rocks resemble arenites and consist chiefly of such allochems as skeletal fragments or ooids glued together by interstitial sparry cement.
- Microcrystalline allochemical rocks are similar to wackes.
- They consist of such allochems as skeletal fragments or ooids floating in or intermixed with fine grained microcrystalline matrix (micrite).
- Micrite is analogous to mudrock. It consists principally of microcrystalline calcium carbonate matrix in which are scattered less than 10% allochems.
- Biolithites are rocks that were cemented together into limestone while the organisms that constitute them were still alive and growing.
- These include reef limestone and stromatolites.
Allochems: Characteristics and Genesis
Allochems are subdivided into (1) skeletal (biogenic) grains and (2) nonskeletal grains.
Skeletal Allochems
- There are many types of skeletal grains.
- Depositional setting dictates paleoecology.
- The specific biological community that exists in any given setting is determined by such factors as water depth, water temperature, salinity, and turbidity.
- Fossil components in limestone (and in sedimentary rocks generally) reject both age and depositional environment.
Nonskeletal Allochems
- Four major types of nonskeletal grains are recognized: (1) various coated grains (ooids, pisoids, and oncoids); (2) peloids; (3) clumped or aggregated grains (lumps, grapestones); and (4) limestone clasts (limeclasts)-that is, fragments of preexisting limestone derived from intrabasinal and extrabasinal sources.
- These clasts are ripped up, transported, redeposited, and confined within limestone strata.
- Coated grains. There are several types of coated calcium carbonate grains. All share a common spherical to subspherical shape, but they vary in size and degree of internal organization.

- Ooids (also called ooliths) have a nucleus around which concentric layers of calcium carbonate are wrapped.
- Coarser coated grains that are identical to ooids in shape and internal organization are called pisoids (or pisolites).

- Oncoids are the same size as ooids or may be much larger, but they are irregularly shaped.

- They contain no obvious nucleus; overlap. Modern ooids form in marine settings at shallow water depths.
- Although ooids can form in water as deep as 15 m, most form at less than 5 m.
- Ooids and pisoids can be biogenic, inorganic, or both.
- They apparently grow by simple accretion as wave, tidal, and storm currents sweep grain nuclei back and forth in shallow marine seawater supersaturated with dissolved calcium and carbonate.
- Peloids. Most peloids are fine-grained (0.1 to 0.5 mm) sand- to silt-sized clasts of microcrystalline carbonate that lack a coherent internal structure.
- The term implies neither size nor mode of formation.
- Most peloids are rounded to subrounded, but they can bespherical, subspherical, ellipsoidal, or irregular in shape.
- Peloids form in shallow marine low-energy platform carbonate settings.
- Many peloids are fecal pellets of waste matter generated by such organisms as fish and shrimp.
- Others are produced by the micritization of otherkinds of allochems: ooids, oncoids, intraclasts (limeclasts), and abraded skeletal grains.

- Grain aggregates. Grain aggregates form when such carbonate particles as ooids and peloids adhere to one another.
- Most are fine- to medium-grained sand-sized masses that are given specific descriptive names: grapestones, lumps, and botryoidal lumps. Interstitial pores are filled with micrite, but most aggregates are bound together by encrusting organisms.

- Limestone clasts. Limestone clasts (or limeclasts) are ripped-up and transported fragments of pre-existing carbonate derived from extrabasinal and intrabasinal sources.
- Extrabasinal limeclasts differ in composition, texture, and overall character from the limey material in which they occur. Intrabasinal clasts are eroded from the same stratigraphic unit, or even the same stratum, in which they are found now.
Orthochems: Characteristics and Genesis
- Micrite is calcium carbonate mud that occurs as matrix in allochem-bearing limestone or by itself as limey mudrock.
- It consists of silt and clay fragments of aragonite (in modern sediment) or calcite (in ancient limestone).
- Micrite is typically translucent under the microscope with a dull brown cast. It is dull and opaque in hand specimen.
- Spar is carbonate cement. Crystals of spar are generally coarser than micrite. Crystal diameters typically range from 0.02 to 0.1 mm.
- Under the microscope, spar is crystal clear without the hazy brownish cast of micrite.
- Neomorphism- the various diagenetic processes of recrystallization and replacement, including changes in mineralogy- is very common in carbonate rocks.
- Micrite is polygenetic and is most commonly formed by the physical disintegration of calcareous green algal masses within which biogenically produced needles of aragonitic are disseminated.
- Micrite also forms when allochems such as peloids and limeclasts disaggregate; it can also be the by-product of mechanical abrasion and bioerosion (organic burrowing and ingestion) of carbonate grains.
- Spar forms as a simple, primary, pore-filling cement or is generated secondarily by recrystallizating micrite.
The Dunham Classification
- The one major aspect of limestones not reflected in Folk's classification is whether the sparry calcite is primary cement or a secondary recrystallization of micrite.
- Since this determination is often hard to make, Folk's classification is much more descriptive and objective.
- Some geologists felt that the classification should also separate primary from secondary sparry limestones.
- The easiest way to do this is to incorporate grain support into the classification.
- Limestones in which all the grains touch others (grain-supported) were originally porous and were later cemented.
- This is primary sparry cement. Limestones with grains floating in spar (not grain-supported) were probably originally composed of grains floating in micrite that has been recrystallized.
- This is secondary spar, a replacement after micrite.
- These features are important in the Dunham classification scheme (Fig. 11.SA).
- The Dunham classification emphasizes limestone texture, especially grain (allochem) packing and the ratio of grains to matrix.
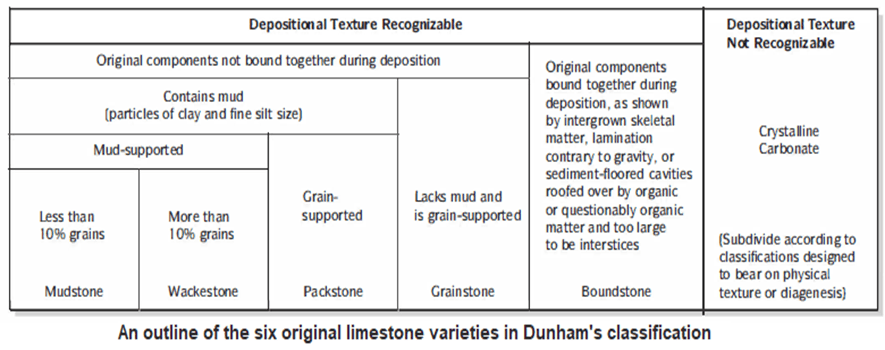


- Five types of limestones were later identified: mudstone, wackestone, packstone, grainstone, and boundstone. All except boundstone accumulate as elastic carbonates; individual components are not bound together during deposition.
- Mudstone, wackestone, and packstone contain mud, which Dunham defined as any silt- or clay sized grains, regardless of composition.
- Mudstone and wackestone are mud-supported. They are limestones in which allochems are scattered through a rock that is basically micrite.
- Packstone contains less mud and is grain-supported.
- Grainstone contains no mud, and allochem grains support one another.
- Limestones in which the components have been bound together from origin (such as reef rocks) are called boundstones.
Diagenetic Environments
- Because seawater is supersaturated with calcium carbonate, it takes just a slight change in water chemistry, or impurities and sites of nucleation, for marine cementation to occur.
- A wide variety of environmental settings (Fig. 11.9) can produce diagenesis in carbonate rocks. In the intertidal zone, for example, there is rapid education of the water chemistry during tides. The result is beach rock, which can form in a matter of years under the right conditions.

- The result of shallow subtidal cementation is a crusty surface known as a hardground. Hardgrounds are often found in ancient limestone and are considered evidence of interruptions in sedimentation, or hiatuses, that may span several thousand years.
- The most extensive areas of early carbonate sedimentation, however, occur above sea level, in zones where fresh meteoric (atmospheric) waters-that is, rain or runoff groundwaterow through the pores of carbonate sediments.
- This area is known as the vadose zone, or zone of aeration above the water table. The phreatic zone, or zone of saturation below the water table, can also produce cementation.
- The most noticeable difference between marine and freshwater cements is that marine cements precipitate high-Mg calcite, whereas meteoric waters can only precipitate low-Mg calcite.
Dolomite and Dolomitization
- Many ancient carbonate rocks consist in whole or in part of the mineral dolomite, CaMg(CO3)2Technically, the name dolostone should be applied to carbonate rock made of the mineral dolomite,
- Dolomitization is the process by which calcium carbonate rock alters to dolostone.
- This process obscures the original texture, which makes classification and genetic interpretation difficult. Where original textures and components survive, limestone classification is used and the degree of dolomitization is noted.
- Dolomite is very similar to other carbonate minerals, so it is not always easy to identify in the field. It is less soluble than calcite and does not readily fizz in dilute acid.
- Dolomite changes the texture of a rock in a noticeable way. For example, dolomitized rocks loo "sugary," and in many instances dolostones have a golden-brown or tan color (in contrast to limestones, which are typically gray).
- In addition, the different solubilities of dolomite and calcite mean that dolostones weather differently.
- Often, the fossils in a limestone will beundolomitized, so that under chemical weathering and dissolution, the dolomitized matrix will remain and there will be voids where calcitic fossils have dissolved away.
- In other cases, dolomite selectively replaces the burrows in a micritic limestone, resulting in a mottled texture.
- Virtually all dolostones are composed of secondary (or replacement) dolomite, meaning that calcite and aragonite form first but are later converted to dolomite.
- This can be proved easily if relict textures of limestone are still visible despite the overprinting of dolomite.
- In some dolostones, there are no relict fossils, ooids, or other evidence of limestone parent material; there are just masses of rhombohedral dolomite crystals.
- Modern settings in which recently formed dolomite occurs are not necessarily sites of primary dolomite.
- Most sites reported as primary are actually locales in which the time interval between the formation of calcium carbonate minerals and their conversion to dolomite is exceedingly short.
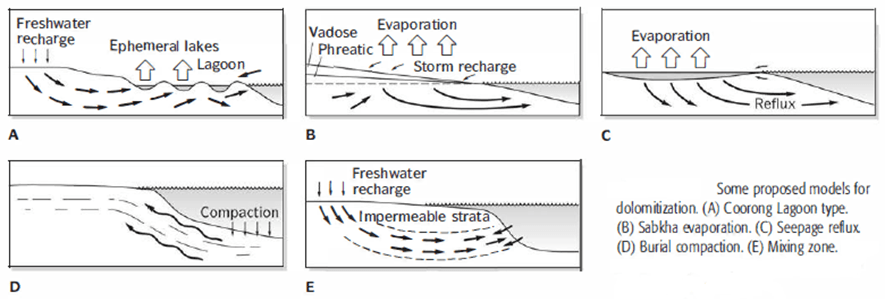
- Figure above summarizes several modern dolomitization sites. Each has a physical-chemical environment that produces dense brines.
- The brines are produced by evaporation until Ca-rich, Mg-poor minerals such as gypsum and anhydrite form.
- Figure A shows one of the best-known modern dolomite sites, Primary dolomitic micrite forms within seepage-fed alkaline lakes and lagoons developed in the interdune rifts.
- Precipitation of gypsum and anhydrite preferentially removes Caz+ and Soi-. The uid that remains is the dolomitizing agent, although high alkalinity and the presence of nearby Mg-rich groundwater (generated during the leaching of subjacent volcanic rocks) may help.
- Dolomite also forms as thin surficial layered patches developed on sabkhas. Figure B shows an example of this setting.
- The complex plumbing system is driven by evaporation. Storms and abnormally high tides food supratidal flats with seawater of normal salinity during the winter and spring.
- During the long, hot, arid summer, net evaporation leads to the crystallization of Ca-rich evaporite minerals.
- Upward evaporative pumping causes these dolomitizing fluids to react with aragonite mud.
- Figure C illustrates the formation of brine reflux dolomite.
- A broad hypersaline basin is shown subjacent to the edge of a carbonate platform.
- Hypersaline brines generated by evaporation from lagoonal water in that basin become heavier, and Mg-rich.
- Such brines eventually become dense enough to displace connate waters, resulting in reflux; that is, they slowly seep downward and are flushed through the permeable carbonate sediment, converting it to dolostone.
- Figure D illustrates burial dolomitization, a model that explains the many dolostones that are not intimately associated with evaporites.
- Burial dolomitization occurs when buried Mg-rich mudrock is compacted and dewatered.
- Expelled fluids rich in magnesium then invade and dolomitize the adjacent limestone.
- The mixing-zone dolomitization model is a model developed to explain ancient dolostones that show no apparent relationship with hypersaline brine and evaporite (Fig. E).
- In this model, seawater of normal salinity and an Mg-to-Ca ratio of 5:2 is mixed with fresh groundwater, producing brackish groundwater near the coastline.
- The seawater ostensibly provides enough additional magnesium to supersaturate the brackish groundwater with dolomite.
- Groundwater circulation is thought to flush the brackish dolomitizing groundwater through subjacent limestone.

Carbonate Environments
- Carbonate sediments are fundamentally different from clastic sediments in many ways
- Whereas terrigenous elastic sediments have been eroded from bedrock and transported to the basin of deposition (allochthonous), most carbonate sediments form chemically or biochemically within the basin of deposition (autochthonous).
- Many carbonate particles undergo little or no hydraulic transport, so the physical sedimentary processes that make cobbles, pebbles, sand, and mud are less important than the composition of the carbonate particle itself, whether precipitated inorganically or formed biogenically.
- Another striking difference between carbonate and elastic sediments is the extremely limited conditions under which carbonates constitute the predominant sediment.
- Clastic sediments are found in virtually every sedimentary environment, both marine and nonmarine, whereas carbonates are common only in clear, warm, shallow, tropical to subtropical seas-the so-called carbonate factory.
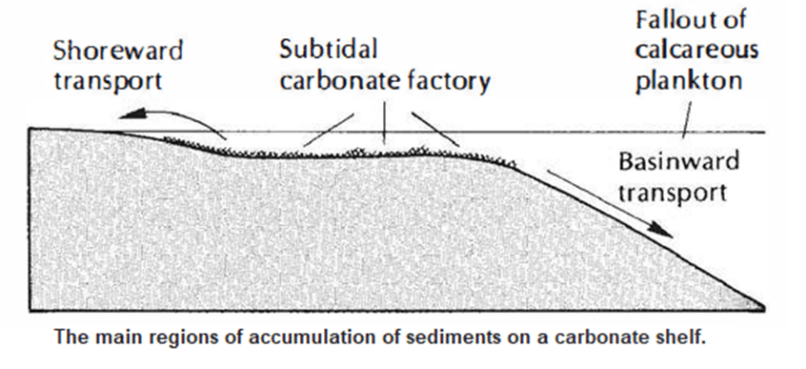
- The environment where carbonates form must meet a number of restrictive conditions that are found only on shallow shelves between 30°N and 30°5 latitudes, because carbonate is produced by environmentally sensitive organisms.
- The shelf waters must be warm-these organisms are tolerant of only a narrow range of temperatures-and shallow-many of these organisms (especially reef-forming corals and lime mud-secreting algae) need light for photosynthesis.
- Most carbonate production takes place in the upper 10 m of seawater and drops off rapidly below this.
- The water chemistry must be normal marine because these organisms are tolerant of a very narrow range of salinity.
- The water must also be clear, free of terrigenous elastics, and only slightly turbulent, because terrigenous quartz sand can abrade the softer calcite, and terrigenous mud can foul gills or digestive tracts, thus hampering the biological activity of most carbonate-secreting organisms.
- If the water is too turbulent, lime mud is stirred up, which cuts down the light and chokes many marine invertebrates.
Peritidal Environments
- Peritidal Environments Because carbonates are so depth-restricted, the position of sea level is a natural boundary between carbonate environments.
- Very different processes occur in peritidal environments and shelf environments; shelf environments are seldom subaerially exposed.
- Salinity and water temperature also undergo extreme fluctuations, which further limits the types of organisms that can live there.
- These chemical conditions have proved to be important for economic reasons as well.
- Peritidal carbonate flats form laterally persistent, evenly bedded limestones and dolostones, which are host rocks for lead and zinc ores and for petroleum.

Diagnostic Features of Peritidal Environments
- Tectonic Setting Like other carbonate shelves, peritidal environments are restricted to clear, shallow, subtropical to tropical marine shelves, which occur in a stable tectonic setting with little relief. These conditions occur primarily in subsiding passive margins or in epeiric seaways.
- Geometry Peritidal environments typically form thin (a few meters thick) but laterally continuous beds representing the various shoreline facies belts, which may build thousands of square kilometers of shelf and form extensive tabular bodies.
- Typical Sequence A shallowing-upward sequence from subtidal lagoon muds or beach sands to intertidal rippled, burrowed, and mudcracked flats to supratidal algal flats (with or without evaporites) and eolian dunes is typical.
- Sedimentology Features of the intertidal and supratidal environment are very distinctive: algal lamination and stromatolites; mud cracks; tidal channel breccias with shells and mudchip rip-up clasts; birdseye texture; dolomitization; and evaporite minerals (especially gypsum, anhydrite, and halite), which may rise diapirically.
- Fossils Stromatolites made by cyanobacteria (blue green algae) and other resistant types are most common, because of the desiccation and the extreme ranges of temperature and salinity. Some mollusks and burrowing worms can survive on the tidal flat, but most shell debris is washed in by storms.
Subtidal Shelf Carbonates
- Modern shallow carbonate banks occur on continental shelves with little or no elastic input from rivers During major epeiric transgressions, however, the continents must have been covered with a few meters of shallow, warm,
- clear water, which produced enormous areas of carbonate banks, reefs, and shoals.
- As long as subsidence is relatively slow, the carbonate- secreting community of organisms can keep up with it by building up substrate fast enough to remain within the photic zone.
- Shelf deposits are similar in many ways to peritidal and reef carbonates, but there are some major differences.
- The water is always normal marine, so the organisms found on the shelf tolerate only a narrow range of salinities.
- Because most of the organisms depend directly or indirectly on light, the depths are shallow, ranging from low-tide level down to 100 or 200 m.
- Like other shallow carbonate environments, carbonate banks form in a narrow range of temperatures (10° to 30°C), so they are found in tropical or subtropical latitudes. Unlike peritidal lagoons, however, the water is always well-oxygenated.

Diagnostic Features of Subtidal Shelf Carbonates
- Tectonic Setting All shelf carbonates require warm, clear, shallow, well-oxygenated, normal marine conditions, which typically exist only on continental shelves or epeiric seas in low latitudes with no significant elastic input.
- Geometry Subtidal shelf carbonates form homogeneous sheets that can cover thousands of square kilometers and reach hundreds of meters in thickness.
- Typical Sequence Under normal conditions, a uniform skeletal pelletal mudstone with remarkable vertical homogeneity is produced. If the sequence shoals upward, the subtidal limestones are capped by intertidal sequences and possibly evaporites.
- Sedimentology Although the mineralogy is almost exclusively aragonite, calcite, and dolomite (with rare shales and evaporites), textures are highly variable.
- Pelletal muds rich in biogenic debris are common, as are ooids, skeletal sands, and bioturbated muds.
- Bedding of variable thickness, with wedge- and lens-shaped units, is particularly characteristic, as are nodular bedding and flaser bedding caused by compaction.
- Fossils The most diagnostic feature is the abundance of fossils of the normal marine fauna, which are tolerant of only a limited range of salinities, light conditions, turbulence, and oxygen content.
- The shallow carbonate shelf supports by far the greatest diversity of marine faunas of any environment on Earth.
Reefs and Buildups
- Carbonate reefs and buildups are completely self-generated.
- They are sediment systems built entirely by the organisms growing in them.
- The term bioherm or biostrome is generally applied to any in situ accumulation of benthic organisms, whether or not it is topographically high or wave-resistant.
- Reefs and build-ups are interesting for many reasons.
- The presence of a reef implies an adequate supply of nutrients, as well as the usual restrictive conditions of carbonate sedimentation, and so provides a valuable indicator of paleoceanographic conditions.
- Reefs also give clues to understanding the paleoenvironment at the times in the past when building up was especially common.
- Buildups also serve as important stratigraphic traps for oil.
- The reef is divided into various regions (sometimes called facies) by environmental conditions.
- In general, most reefs can be divided into three distinct facies: the reef core, the reef flank, and the interreef.
- The reef core is massive, unbedded, and composed of the framework builders filled in by an interstitial matrix of lime mud and skeletal sand.
- The crest of the reef core is subject to the greatest wave energy, so the growth forms are low and encrusting.
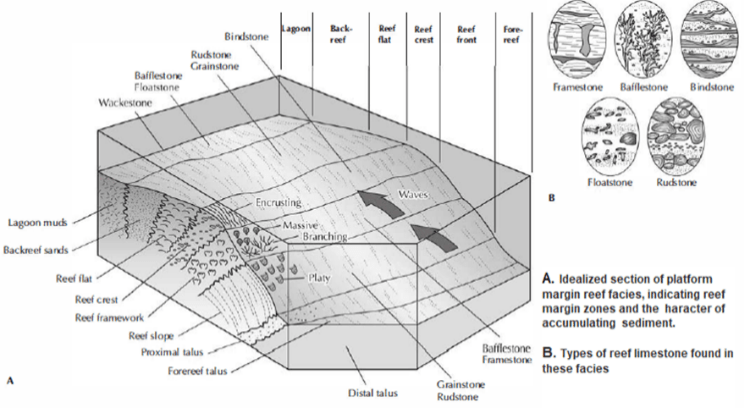
- The upper part of the reef front is also made of encrusting and massive framework organisms.
- Behind the reef crest is the reef flat, which is subjected to considerable current and wave activity but is usually sheltered from the worst pounding of the waves.
- Because the water is shallow, the framework builders grow rapidly, and form irregular clumps and patches separated by current work channels of sand and rubble.
- The most sheltered area of all is the backreef which experiences waves only during storms.
- It is covered with carbonate sand derived from the biological breakup of carbonate organisms and is moved around by storm waves.

- Below the growing part of the reef core is the reef bank. Debris from the reef washes or slumps down the reef front and into the flank beds, which dip gently away from the core.
- As a result, the flank beds are usually composed of bedded limestone conglomerates made of reef debris and lime sands that have washed in during storms.
- The interreef facies deposits are usually composed of thin-bedded, pelletal limemuds that are rich in skeletal sands.
- Often, the only indication of an interreef location is the rare presence of debris from the reef itself, which has been brought there by extreme storm waves.
- In basins with restricted circulation, evaporite minerals are common in the interreef facies.
Diagnostic Features of Reefs and Buildups
- Tectonic Setting Reefs and buildups form at the edges of carbonate banks where upwelling from deeper waters brings up nutrients.
- They are extremely limited by depth, temperature, salinity, and nutrients. Like other carbonate bank deposits, buildups are found in shallow, low-latitude, passive margins or epeiric seas free of elastic input.
- Geometry Small, local mound like or bank like accumulations occur that show rapid lateral changes in fades and thickness form.
- Patch reefs may be only a few meters high and wide, but some large reef complexes are hundreds of meters thick and kilometers wide.
- Typical Sequence Other than reef succession, there is no typical stratigraphic sequence of reefs and buildups.
- The lateral relationships of the various reef facies are more important than the vertical pattern, although both factors may work in concert to form a complex of massive reefs and bedded interreef limestones.
- Sedimentology Framework builders are dominant, so an entire deposit grows and is bound together in situ. Interstitial lime mud, skeletal fragments, and reef-rock breccias are formed in crevices between the framework organisms.
- Reefs are formed exclusively of calcite or aragonite, although the reef core is highly susceptible to dolomitization.
- The back-reef flats may become evaporitic in the appropriate environment.
- Fossils Reefs are formed almost entirely of characteristic fossils whose ecology determines the growth and shape of the reef.
- Reef organisms are extremely sensitive to temperature, salinity, light, and terrigenous mud, so they are excellent indicators of environmental conditions.