Metamorphism of Arenaceous, Argillaceous and Basic Rocks
13.1 Metamorphism of mafic rocks
- Mafic rocks include volcanics from basaltic to andesitic composition, gabbros and diorites, and immature mafic graywackes.
- Metamorphosed volcanics are by far the most common, and the volcanic parent rocks occur in a wide variety of tectonic settings, from oceanic crust to volcanic arcs to rifts.
- Hydrous minerals are not common in high-temperature mafic igneous rocks, so hydration is a prerequisite for the development of the metamorphic mineral assemblages that characterize most facies.
- Unless H2O is available, mafic igneous rocks will remain largely unaffected in metamorphic terranes.
- Coarse-grained intrusives are the least permeable and thus most likely to resist metamorphic changes, whereas tuffs and graywackes are the most susceptible.
- Mafic rocks are chemically complex and variable. Because most reactions are continuous reactions, the grade at which a particular mineral appears or disappears during prograde metamorphism may vary from one rock or location to another.
- Most of the common minerals in mafic rocks exhibit extensive solid solution.
- Because these minerals can accommodate a lot of chemical diversity, metabasites tend to have fewer phases than pelites. This results in fewer reactions and isograds.
- The principal mineral changes in metabasalts are due to the breakdown of the two most common basalt minerals: plagioclase and clinopyroxene.
- As temperature is lowered, the more Ca-rich plagioclases become progressively unstable.
- There is thus a general correlation between temperature and the maximum An-content of the stable plagioclase.
- Clinopyroxene breaks down to a number of mafic minerals, depending on grade. These minerals include chlorite, amphibole, epidote, a metamorphic pyroxene, etc.
Mafic Assemblages at Low Grades
- Very-low-grade metabasites are usually only partly altered to very-fine-grained and messy-looking minerals.
- At the lowest grades of the zeolite facies, volcanic glass is altered to the zeolites heulandite or stilbite along with phyllosilicates such as celadonite, smectite, kaolinite, or montmorillonite, plus secondary quartz and carbonate.
- At slightly greater depths, albite replaces more calcic detrital igneous plagioclase, chlorite appears, and heulandite is replaced by laumontite
- The first appearance of laumontite occurs before heulandite or analcime are totally lost, and these zeolites may coexist over an interval.
- Although prehnite and pumpellyite are stable in the upper zeolite facies, eventually laumontite is lost with increasing grade, and prehnite pumpellyite quartz becomes stable.
- This transition marks the beginning of the prehnite–pumpellyite facies at a temperature of about and a depth of 3 to 13 km.
- Prehnite and pumpellyite disappear at higher grades. Prehnite generally disappears first, giving way to actinolite.
- ACF diagrams with representative mineral assemblages that develop in metamorphosed mafic rocks in the zeolite and prehnite–pumpellyite facies are shown in Figure 5.
- Most mafic rock compositions (shaded area) in the zeolite facies develop a mineral assemblage such as chlorite heulandite (or laumontite) calcite quartz albite.
- Kaolinite develops in more aluminous rocks. The transition from zeolite to prehnite–pumpellyite facies is marked by the loss of huelandite and laumontite, and the development of prehnite, pumpellyite, and (at a higher grade) actinolite.
- The crossing tie-lines in Figure 5b are a result of combining both the lower-grade prehnite-bearing zone with the higher-grade pumpellyite–actinolite zone on the same diagram.
- Thus, we might expect chlorite + prehnite + calcite in the lower prehnite–pumpellyite facies, and pumpellyite + actinolite + chlorite in the upper part.
- Pumpellyite disappears as rocks enter the greenschist facies, where it is replaced by actinolite + epidote at ~270 to 300℃.
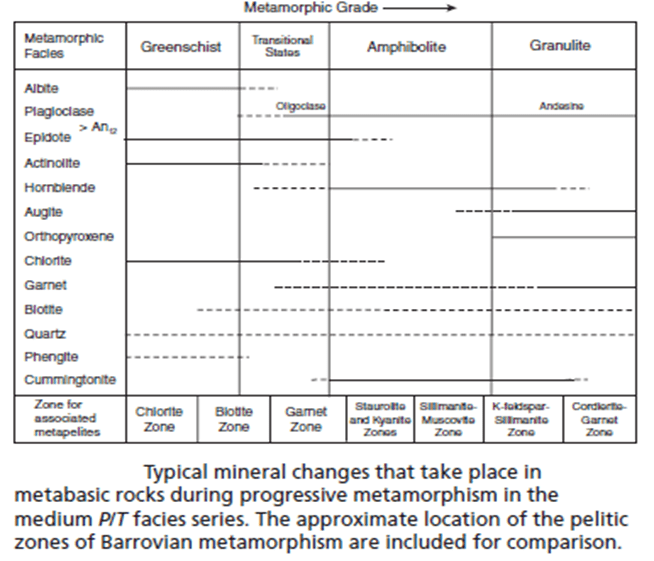
- Figure 6 summarizes the mineral changes that typically occur in metabasites in the zeolite, prehnite–pumpellyite, and lowest greenschist facies.
- Different mineral stability ranges characterize the rocks throughout various hydrothermal fields and burial metamorphic terranes as a result of variations in composition, fluids, and local geothermal gradient.

Mafic Assemblages of the Medium P/T Series: Greenschist, Amphibolite, and Granulite Facies.
- The greenschist, amphibolite and granulite facies constitute the most common facies series of regional metamorphism.
- The zeolite and prehnite–pumpellyite facies are not represented in the Scottish Highlands, and metamorphism of mafic rocks is first evident in the greenschist facies, which correlates with the chlorite and biotite zones of the associated pelitic rocks.
- The mineralogy reflects re-equilibration to metamorphic conditions.
- Typical metabasic minerals include chlorite, albite, actinolite, epidote, quartz, and, in some cases, calcite, phengite or biotite, or stilpnomelane.
- The predominance of chlorite, actinolite, and epidote impart the green color from which the mafic rocks and the facies get their name.
- Figure 7 is an ACF diagram representing typical metamorphic mineral assemblages in metabasites of the greenschist facies.

- The most characteristic mineral assemblage is chlorite + albite + epidote + actinolite ± quartz. Chloritoid is not recognized in the classical Barrovian sequence.
- The transition from greenschist to amphibolite facies in mafic rocks involves two major mineralogical changes.
- The first is the transition from albite to oligoclase. The second is the transition from actinolite to hornblende as amphibole becomes able to accept increasing amounts of aluminium and alkalis at higher temperatures.
- The reactions that generate calcic plagioclase and hornblende are complex and continuous, involving the breakdown of epidote and chlorite to supply the Ca and Al for the anorthite and Al-hornblende components
Ab + Ep + Chl + Qtz = oligoclase + tschermakite + Mt + H2O
- Tschermakite, an Al-rich component in the amphibole.
- Metabasites in the amphibolite facies are characterized by the assemblage hornblende + plagioclase.
- Figure 8 is an ACF diagram illustrating the typical assemblages. Because of hornblende solid solution, much of the compositional area for mafic rocks lies in the two-phase Hbl-Plag field.
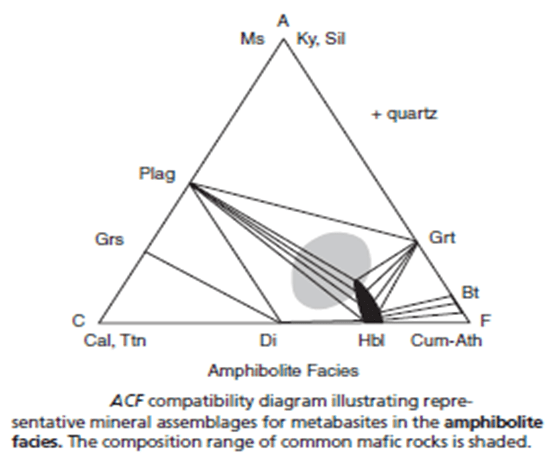
- In many cases, hornblende becomes more brown and less green in thin section as grade increases. The brown color is attributed to higher Ti content.
- ACF compatibility diagram illustrating representative mineral assemblages for metabasites in the amphibolite facies. The composition range of common mafic rocks is shaded.
- The transition from amphibolite to granulite facies occurs in the range 650 to 850℃.
- The granulite facies is characterized by the presence of a largely anhydrous mineral assemblage.
- In metabasites the critical mineral assemblage is orthopyroxene clinopyroxene plagioclase quartz. Garnet is also common, Figure 9 is an ACF diagram for typical mineralogy in granulite facies metabasites.
- The origin of granulite facies rocks is complex and controversial. There is general agreement, however, on two points:
- Granulites represent unusually hot crustal conditions. Granulite facies rocks represent temperatures in excess of 700℃.
- Granulites are dry. The only reason these rocks didn’t melt on a large scale was the lack of available H2O.
- The granulite facies encompasses a wide range of pressure, giving way to the eclogite facies when the pressure reaches the point that plagioclase is no longer stable.

Mafic Assemblages of the Low P/T Series: Albite–Epidote Hornfels, Hornblende Hornfels, Pyroxene Hornfels, and Sanidinite Facies
- The mineralogy of metabasites in the low-pressure facies is not appreciably different from that in the medium-pressure facies series the albite–epidote hornfels facies correlates with the greenschist facies into which it grades with increasing pressure.
- Similarly, the hornblende hornfels facies correlates with the amphibolite facies, and the pyroxene hornfels correlates with the granulite facies
- At low pressure, the albite–oligoclase transition occurs and it marks the end of the albite–epidote hornfels facies. A transitional zone typically separates it from the hornblende hornfels facies in contact aureoles.
- Ca-poor amphiboles, such as cummingtonite, are more widespread at lower pressure.
- Sanidinite facies is not evident in basic rocks.
- Mafic Assemblages of the High P/T Series: Blueschist and Eclogite Facies
- In contrast to low P/T metamorphism is the mafic rocks, that develop conspicuous and definitive mineral assemblages under high P/T conditions.
- Most mafic blueschists are easily recognizable by their color, and are useful indicators of ancient subduction zones.
- The great density of eclogites suggests that subducted basaltic oceanic crust becomes denser than the surrounding mantle.
- Between the zeolite facies and blueschist facies a transitional zone may occur, characterized by the high-pressure phase lawsonite.
- Alternative paths to the blueschist facies depend on the local geothermal gradient and may follow paths such as zeolite facies →prehnite–pumpellyite facies →blueschist facies, or (zeolite facies) →(prehnite–pumpellyite facies) → greenschist facies →blueschist facies.
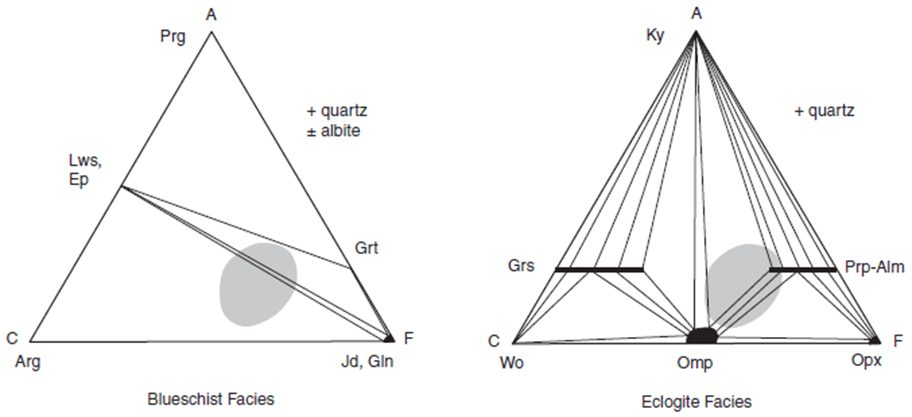
- The blueschist facies is characterized in metabasites by the presence of a sodic blue amphibole (notably high in glaucophane component), stable only at high pressures.
- The association glaucophane lawsonite is diagnostic. Several reactions can generate glaucophane and define the transition to the blueschist facies.
- Examples are: Tr + Chl + Ab = Gln + Lws
- All are pressure sensitive.
- Albite breaks down at high pressure by reaction to jadeitic + quartz.
NaAlSi3O8 = NaAlSi2O6 +SiO2
- The assemblage jadeite quartz indicates high pressure blueschist facies (see fig).
- Low-pressure blueschist facies can be distinguished by the presence of jadeite without quartz.
- The Sanbagawa belt of Japan represents the low-pressure type of blueschist facies, whereas the high-pressure type occurs in the Franciscan Formation of western California (Greece), (western Alps).
- Lawsonite remains stable throughout most of the blueschist facies and even into the eclogite facies.
- The transition to the eclogite facies is marked by the development of the characteristic mafic assemblage omphacitic pyroxene almandine– pyrope–grossular garnet, creating dense, beautiful green and red rocks.
- The eclogite facies (see fig) represents pressures at which plagioclase is no longer stable.
- The albite component of plagioclase breaks down in the blueschist facies.
- ACF compatibility diagram illustrating repre - sentative mineral assemblages for metabasites in the blueschist facies. The composition range of common mafic rocks is shaded.
- ACF compatibility diagram illustrating representative mineral assemblages for metabasites in the eclogite facies. The composition range of common mafic rocks is shaded.
- Blueschist facies to eclogite facies transition involves glaucophane and paragonite reacting to form the garnet– omphacite assemblage:
Gln + Pg = Prp + Jd + Qtz + H2O
- Along higher geothermal gradients, the amphibolite facies, or even the granulite facies, may give way at high pressure to the eclogite facies as well.
- At higher temperature, plagioclase reacts with amphibole, orthopyroxene, clinopyroxene, and/or olivine via a number of possible reactions to form omphacitic clinopyroxene pyrope–grossular garnet as pressure increases.
- Any of the plagioclase breakdown reactions mark the transition from amphibolite or granulite facies to eclogite facies.
- Na, Ca, and Al, rather than residing in plagioclase, are then ensconced in the pyroxene and garnet in eclogites.
Pressure–Temperature–Time (p-t-t) paths
- The concept of metamorphic facies series suggests that a traverse upgrade through a metamorphic terrane following a metamorphic field gradient may cross through a sequence of facies. are the temporal and spatial mineralogical changes in an area the same?
- Let’s consider the complete set of T-P conditions that a rock now at the surface may experience during a metamorphic cycle from burial to metamorphism (and orogeny) through uplift and erosion.
- Such a cycle, called a pressure– temperature–time path, or P-T-t path, applies to conditions experienced by progressively subducted crust and convecting mantle wedge materials as they moved through the subduction zone complex.
- The following figure illustrates some examples of modeled P-T-t paths representing common types of metamorphism.
- The paths illustrated are schematic, and numerous variations are possible, depending upon the style of deformation and the rates of thickening, heat transfer, magmatism, and erosion, etc. that are assumed.

- Path (a) in the figure is considered to be a typical P-T-t path for an orogenic belt experiencing crustal thickening.
- During the thickening stage the pressure increases much more rapidly than the temperature because of the time lag required for heat transfer (pressure equilibrates nearly instantaneously, but heat conducts very slowly through rocks).
- A rock in the thickened crustal block thus quickly approaches its maximum depth (Pmax)while remaining relatively cool.
- Although the exact shape, size, and position of an orogenic P-T-t path such as path (a) may vary with the constraints of the model, most examples of crustal thickening have the same general looping shape.
- Paths such as (a) are called “clockwise” P-T-t paths in the literature and are considered to be the norm for regional metamorphism.
- Path (b) in the figure represents a different situation in which a rock is heated and cooled at virtually constant pressure by magmatic intrusion at shallow levels.
- This may be an appropriate P-T-t path for contact metamorphism. Depending upon the extent of magmatic activity and its contribution to the crustal mass, any number of paths transitional between (a) and (b) can be imagined, representing a gradation from highpressure blueschist metamorphism to (Barrovian) regional.
- Metamorphism to “regional contact metamorphism” with numerous plutons to local contact metamorphism.
- Path (c) is an example of what is conventionally called a “counterclockwise” P-T-t path.
- This commonly occurs in high-grade gneisses and granulite-facies terranes and is believed to result from the intrusion of relatively large quantities of magma into the lower and middle crust.
- The rapid introduction of magmatic heat and mass causes both the pressure and temperature to increase in unison below the intrusions.
- This is followed by nearly isobaric cooling because the high density of the mafic magma does not lead to crustal buoyancy, so that uplift and erosion are limited.
- “Counterclockwise” P-T-t paths are not restricted to granulite terranes. The behavior has been shown to occur in other settings, usually associated with high magmatic heat flow.
- Although the paths that rocks are believed to follow during metamorphism are called pressure–temperature–time paths, time is not quantitatively illustrated on the P-T diagrams produced.
13.2 Metamorphism of argillaceous rocks
- Pelitic rocks are derived from clay-rich sediments and are of particular importance in studies of metamorphism because they develop a wide range of distinctive minerals.
Representation of Pelite Assemblages on Phase Diagrams
- In chemical terms, reactions in pelitic rocks principally involve the components Si02, Al 20 3, FeO, MgO, K20 and H20, and most theoretical and experimental studies attempt to model natural rocks using this simplified system, which has become known as the KMF ASH system from the initial letters.
- Other components, especially Fe20 3, Ti02, MnO, CaO, Na20 and C may also be present to a significant extent, but with some important exceptions they do not usually play a major role in the reactions that produce the key metamorphic index minerals.
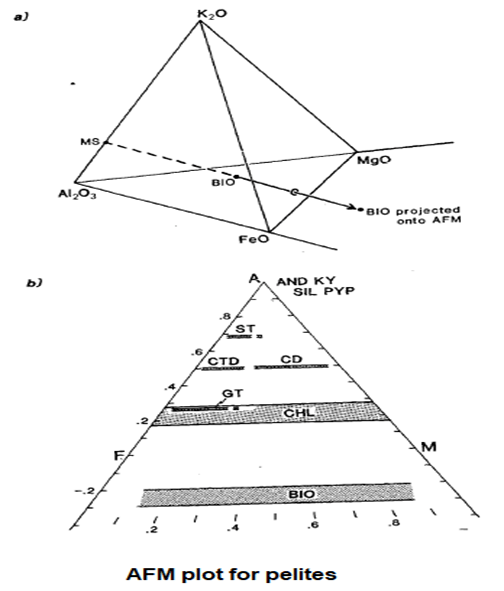
- Eskola, who pioneered the representation of metamorphic ". phase diagrams, attempted to solve the problem of producing a manageable triangular a diagram for pelites by combining FeO and MgO as a single component to produce a manageable triangular AKF diagram with A12O3,K2O and FeO + MgO as the corner components.
- A solution to the problem is provided by the AFM projection. This projection is based on the fact that most metapelites contain muscovite and involves projecting from the composition of muscovite on to the AL2O3-FeO-MgO (AFM) face of the Al2O3-K2O-FeO-MgO (AKFM) tetrahedron, in addition to projecting from quartz and water.
- This procedure is illustrated in fig., which shows a biotite composition projected on to the AFM face by drawing a line from muscovite through biotite until it intersects the AFM plane.
- Note that for biotite, the projected composition on the AFM diagram lies outside the original AKFM tetrahedron.
- Hence the AFM coordinates of any analysis of an AKFM mineral for plotting on the AFM projection are given by:
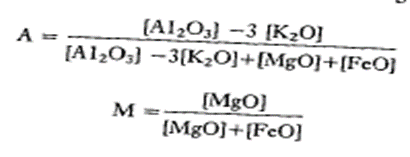
Pelitic Rocks at Low Grades
- Clay-rich sediments may undergo extensive changes during diagenesis, and there is nu sharp distinction between diagenetic and metamorphic processes.
- During the advanced stages of diagenesis many clays become unstable and pelitic sediments are converted to mixtures of chlorite and illite, with some of the kaolinite group minerals possibly also present.
- Some authors use the term anchizone for the zone of chlorite-illite rocks transitional into metamorphism, and the term epizone for the succeeding lowest grades of metamorphism in which illite is replaced by white mica.
- The very fine white micas of epizonal slaty rocks are known as sericite and the dominant constituent is phengite, a variety of muscovite.
- It is not feasible to map conventional isograds in very low grade pelites, but two techniques have been developed to study the variations in grade.
- Both depend on the progressive recrystallisation that occurs during heating. One method is to use X-ray diffraction to measure the crystallinity of illite, grades.
- The other method is to measure the reflectance of original organic matter on polished surfaces, since this also increases with recrystallisation to graphite.
Metamorphism of Pelite in the Barrovian Zonal Scheme
- The classic scheme found in the Scottish Highlands provide excellent example of the way in which mineralogy of pelites can vary with increasing P and T.
- Assemblages are summarised in the table.

Chlorite zone
- In the chlorite zone of Scotland, pelitic rocks are fine grained slates, often with graphite.
- The fine grain size makes it difficult to study these rocks under microscope but they typically contain chlorite and phengitic muscovite wit vanable amounts of quartz, albite, and accessories such as pyrite. Some pelites may contain K-feldspar, stilpnomelane or minor calcite.
- In some regions, chlorite zone rocks are coarse grained schists and the minerals are more readily identified.
Biotite zone
- The rocks in which biotite develops are not strictly pelites but are greywackes with detrital K-feldspar, in which biotite can form by the reaction:
K-feldspar + chlorite → biotite + muscovite + quartz + H2O
- True pelites lack K-feldspar, and in them biotite is formed at somewhat higher temperatures through another continuous reaction:
phengite + chlorite > biotite + phengite-poor muscovite + quartz + H2O.
- The temperature at which this reaction commences depends on the extent of phengite substitution in the initial mica, and the XMg values of the chlorite and mica.
- High phengite content and low XMg favour biotite growth at lower temperature. The association chlorite + muscovite + biotite is stable over a wide temperature interval, emphasising the markedly continuous nature of this reaction.
Garnet zone
- The next mineral to appear in the Barrovian sequence is garnet, and at this grade pelitic rocks are typically schists whose minerals are coarse enough to be readily identified in thin section, even though the actual grain size and textures may vary between different areas.
- The garnet isograd is usually easy to trace in the field because garnet appears in a wide range of rock composition as well as in pelites at about the same grade.
- Frequently it forms conspicuous porphyroblasts and this helps to distinguish almandine-rich garnets typical of the garnet zone.
- A typical garnet zone pelitic rock has the assemblage:
garnet + biotite + chlorite + muscovite + quartz + plagioclase (albite)
- The characteristic garnet of the garnet zone is rich in almandine and probably grows by a continuous reaction such as:
chlorite + muscovite--> garnet + biotite + quartz + H2O proceeds.
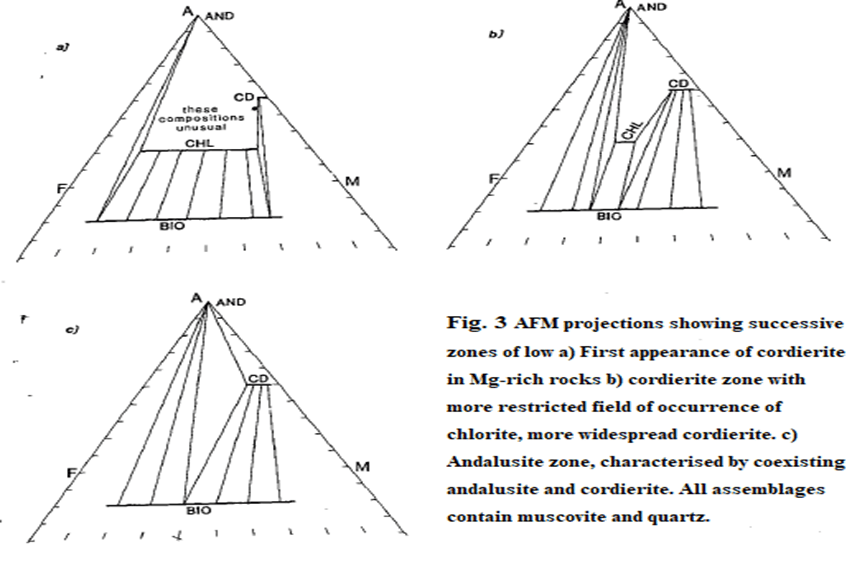
- These continuous changes can be demonstrated conveniently by means of a series of AFM diagrams, shown in figure.
- The biotite zone assemblage muscovite + quartz+ biotite + chlorite with its three degrees of freedom is represented in figure by lines representing a range of chlorite compositions that can coexist with biotite, and a range of biotite compositions that coexist with chlorite. and is represented in figure. by tie-lines connecting specific chlorite and biotite compositions.
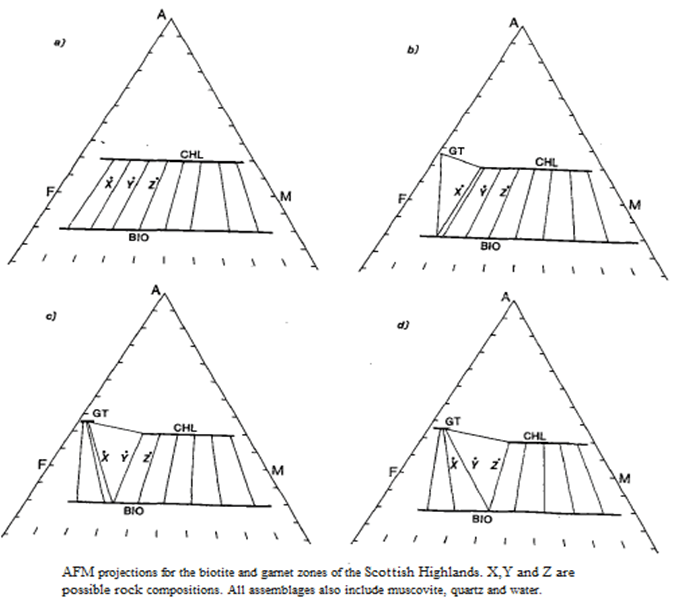
- The typical garnet zone assemblage, muscovite + quartz + chlorite + biotite + garnet is shown in (b)-(d) as a triangle whose comers represent the unique compositions of the phases that coexist at the P-T conditions for which the diagram is drawn.
- Because garnet has a marked preference for Fe, it develops first in the most Fe-rich rocks fig (b).
- With heating, the continuous reaction (reaction [3.3]) progresses, further restricting the range of possible chlorite compositions.
- Points X, Y and Z in Fig represent the compositions of different pelitic rocks.
- At the P-T conditions of Fig. (b), only rock X will develop garnet, whereas by the higher temperature conditions represented by Fig. (d), all three rocks contain garnet and in rock X all chlorite has been consumed and the rock has the assemblage muscovite + quartz + garnet + biotite.
- Figure (c) represents an intermediate temperature. The significance of Fig is two-fold; it shows schematically how the compositions of chlorite, biotite and garnet evolve with temperature in the garnet zone, with Fe-rich chlorites becoming unstable.
- secondly it demonstrates the influence of bulk rock composition on the precise temperature at which index minerals appear.
- In some true pelites chloritoid occurs in the garnet zone.
- It is more commonly present in the assemblages garnet + chloritoid + chlorite + muscovite + plagioclase + quartz or chloritoid. + chlorite + muscovite + paragonite + plagioclase + quartz.
- Figure (a) is an AFM diagram showing a full range of garnet zone assemblages including those with chloritoid.
- The garnet zone is also often marked by a change in plagioclase compositions.
- At lower grades the only plagioclase found is albite, and Ca may be present in the rock in accessory epidote or other phases. Within the garnet zone oligoclase or andesine appear.
Staurolite zone
- It is only in this zone that the distinction between 'true pelites' and other mica schists becomes apparent because staurolite grows in a range of Al-rich, Ca-poor pelitic rocks.
- Many garnet-mica schists prove to be of unsuitable composition for staurolite to form and should mostly be referred to as semi-pelites.
- A critical factor in this is the stabilisation of plagioclase at this grade, which allows available Ca (e.g. in epidote) to combine with additional Al to produce anorthite component.
- The effect is analogous to moving the rock analysis downwards on the AFM diagram at constant 'M' value, to lie in the garnet-biotite field (Fig. 2(b)).
- Staurolite zone pelites contain the assemblage staurolite + garnet+ biotite + muscovite + quartz + plagioclase.
- In areas where suitably aluminous pelites occur, staurolite is produced from breakdown of chloritoid by a reaction such as: chloritoid + quartz --> staurolite + garnet + H20
- Textural evidence for a reaction of this type is found in some staurolite schists that contain relics of chloritoid preserved as inclusions in garnet.
- Staurolite is also found in pelitic rocks that are of unsuitable composition to have ever developed chloritoid staurolite may have been produced by the discontinuous reaction: garnet + muscovite + chlorite --> staurolite + biotite + quartz + H20.
- The difference between Figs 2(a) and 2(b) is the replacement of the garnet-chlorite tie-line with a staurolite-biotite tie-line. As a result, staurolite is found in a wider range of rocks than chloritoid.
- Staurolite may be produced by continuous reaction involving the remaining phases, for example: chlorite + muscovite --> staurolite + biotite + quartz + H2O
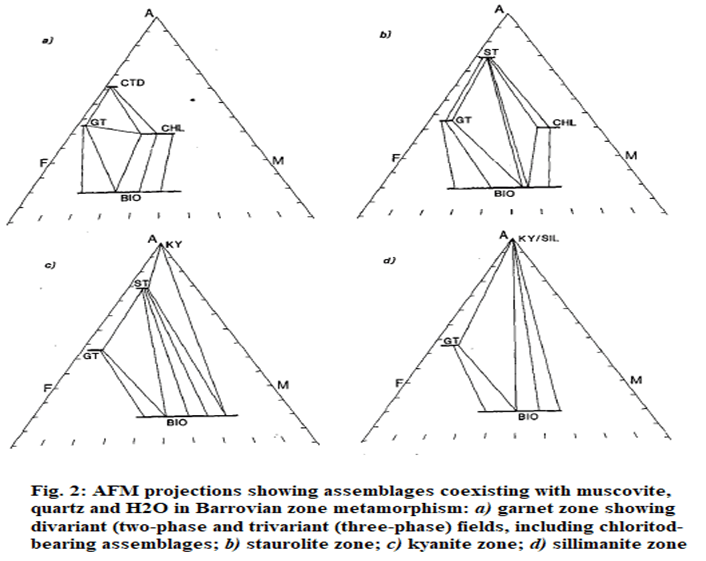
- The difference between Figs 2(a) and 2(b) is the replacement of the garnet-chlorite tie-line with a staurolite-biotite tie-line. As a result, staurolite is found in a wider range of rocks than chloritoid.
- Staurolite may be produced by continuous reaction involving the remaining phases, for example: chlorite + muscovite --> staurolite + biotite + quartz + H2O
Kyanite zone
- The Barrovian kyanite zone is typified by a range of assemblages including the staurolite zone assemblage of garnet + staurolite + biotite (+ muscovite + quartz) as well as those with kyanite: kyanite + staurolite + biotite or kyanite + biotite (+ muscovite + quartz).
- These assemblages are illustrated in Fig.2(c), which differs from Fig.2(b) in that the staurolite-chlorite tie-line is replaced by a kyanite-biotite tie-line.
- This change corresponds to a discontinuous reaction:
muscovite + staurolite + chlorite → biotite + kyanite + quartz + H2O
- This reaction can only take place in pelites with relatively Mg-rich minerals.
- Further growth ofkyanite can occur by the continuous reaction:
staurolite + muscovite + quartz--> Al2SiO3 + biotite + H2O
- This has the effect on the AFM diagram of enlarging the kyanite-biotite field.
Sillimanite zone
- This zone differs from the kyanite zone only in the presence of sillimanite, and kyamte may also still be present.
- The sillimanite normally occurs in the form of very fine needles which may be matted together or penetrate grains of biotite or quartz and is known as fibrolite.
- The reaction that represents the transition from the kyanite zone is the polymorphic transition:
kyanite → sillimanite
- However, the fact that some kyanite commonly remains, suggests that this reaction is very sluggish.
- Instead, it is probable that much of the sillimanite is produced from breakdown other minerals.
- For example, the reaction
staurolite + muscovite + quartz--> Al2SiO3 + biotite + H2O
- appears to take place over a range of temperatures spanning the boundary between the stability fields of kyanite and sillimanite zone.
- Staurolite disappears from muscovite-quartz petites as a result of the discontinuous reaction
staurolite +muscovite+ quartz-> garnet+ biotite + sillimanite + H2O
- This causes staurolite to disappear from the AFM diagram.
Variations on the Barrovian Zonal Pattern
- However, it is not unusual for pelitic rocks to develop different and often equally distinctive mineral assemblages, reflecting different P-T conditions of metamorphism. There are three general ways in which different assemblages and zones may be produced:
- Metamorphism continues to still higher temperatures so that additional zones are present.
- Metamorphism takes place at lower pressures.
- Metamorphism takes place at higher pressures.
High Temperature Metamorphism of Pelites
- In some metamorphic belts the sillimanite zone is succeeded by higher grade zones in which the rocks are often migmatites.
- This means that they are literally 'mixed rocks' usually predominantly schists but with pods, veins or layers of leucocratic material of broadly granitic composition
High Temperature Phase Equilibria in Pelites
- The study of high temperature reactions in pelites is made are complex by the development of a melt phase whose composition is not fixed, and indeed the possible range of compositions is not well known.
- Several theoretical studies have been made that predict sequence of zones that may develop.
Upper silimanite zone:
- The best known isograd above the first incoming of sillimanite is the so-called second sillimanite isograd which represents the growth of additional sillimanite from the breakdown of muscovite:
muscovite + quartz---> A12Si0s + K-feldspar + H2O
- Hence the upper sillimanite zone is characterised by the coexistence of sillimanite and K-feldspar rather than by only one mineral.
- The second sillimanite isograd is a particularly useful indicator of metamorphic grade because it develops in a very wide range of rock types.
- In these areas migmatite features are first developed close the second sillimanite isograd and therefore muscovite breakdown may involve a melt phase also.
- At the moderate pressures of Barrovian metamorphism, this reaction takes place:
muscovite + biotite + quartz + H2O + sillimanite + melt
- This reaction involve an aqueous fluid phase which dissolves in the melt produced. The melting reaction will therefore cease when all the pore water has been assimilated in the melt.
- Further melting can occur through the breakdown of hydrous minerals, which liberate water that is then dissolved in the melt:
muscovite + quartz+ K-feldspar + sillimanite + melt.
- The sequence of above reactions would give rise to the development of migmatites a little below the second sillimanite isograd, which is defined by the appearance of sillimanite + K-feldspar.
Cordierite-garnet-K-feldspar zone:
- At still higher grades, pelitic rocks develop assemblages .with cordierite, garnet, K-feldspar and sillimanite, though not all these necessarily occur together.
- The assemblages result from continuous reactions such as:
biotite + sillimanite + quartz~ K-feldspar + cordierite + melt
biotire + sillimanite + quartz~ K-feldspar + garnet + melt
- Whether cordierite or garnet develops depends partly on pressure (cordierite is favoured by lower pressures, garnet by higher pressures) and partly on the Mg/Fe ratio of the ruck (garnet will form in Fe-rich rocks, cordierite in Mg-rich ones).
- Above reactions lead to melting in Mg-rich and Fe-rich compositions respectively due to dehydration breakdown of biotite, but melting only takes place in the full range of biotite sillimanite schists when the temperature for the discontinuous reaction is attained:
biotite + sillimanite + quartz-> cordierite + garnet + K-feldspar + melt
- The garnet-cordierite-K-feldspar assemblage is typical of high grade pelitic migmatites, and often taken to mark the beginning of the granulite facies; however' even higher grade assemblages are sometimes found.
Ultra-high grade zones:
- High grade granulite facies rocks contain orthopyroxene.
- At medium pressures, very high temperatures are necessary to permit orthopyroxene to but the formation of orthopyroxene is also pressure dependent and at higher pressures this phase is stable to lower temperatures. Orthopyroxene assemblages can be related to the more common cordierite-garnet assemblages through the equilibrium:
Al2SiO5 + orthopyroxene = cordierite + garnet
- with sillimanite + orthopyroxene stable at higher temperatures than cordierite + garnet. Orthoamphiboles are also sometimes found in high grade pelitic rocks. In a few part of the world
- The stable association sapphirine + quartz has been found. This assemblage is stable at even higher temperatures than sillimanite + quartz has been found and in found in rocks that totally lack hydrous minerals, except as products of retrogression
- It may be formed on heating through the reaction:
sillimanite + orthopyroxene à sapphirine + quartz
- This is probably the highest grade assemblage found developed on a region scale in metasediments, requiring temperatures of at least 850°C under even hotter conditions, up to 1000 °C
Metamorphism of pelites at low pressures
- Pelitic rocks may be heated over a comparable range of temperatures to the Barrovian zones but at lower pressures (i.e. nearer the surface) both in contact metamorphism and in some types of regional metamorphism involving high heat flow.
- The principal differences from the Barrovian zonal scheme may be summarised as follows:
- Kyanite. does not occur, but andalusite may be present.
- Cordierite is more common and forms at lower temperatures.
- Garnet is less abundant or absent, and staurolite may also be lacking.
- Migmatites are not developed until above the second sillimanite isograd.
- A variety of different zonal patterns have been desc1ibed from relatively low pressureme amorphism, reflecting the range of pressure-temperature ratios that is possible.
- Their Buchan (D) zonal sequence provides a good example of distinctly low pressure metamorphism fairly typical of many thermal aureoles.
Biotite zone
- The lowest grade rocks in this area are similar to those of the Barrovian biotite zone, having the assemblage biotite + chlorite + muscovite + quartz.
Cordierite zone
- Cordierite appears as the first distinctive index mineral through the continuous reaction:
- chlorite + muscovite -> cordierite + biotite + quartz + H2O
- This reaction, which is probably also responsible for the production of cordierite 'spots' in many 'spotted slates' is apparently very similar to reaction that gives rise to the garnet isograd
- Cordierite first develops in rocks with high XMg values, becoming more common with increasing temperature as the continuous reaction progresses, expanding the field of cordierite + chlorite + biotite on the AFM projection.
- Andalusite can occur in this zone, in relatively Fe-rich rocks, and coexists with biotite but not
Andalusite zone
- Andalusite becomes a possible phase in most pelite compositions at low pressures as a result of the discontinuous reaction:
chlorite + muscovite + quartz -> cordierite + andalusite + biotite + H2O
- Which represents the upper stability limit of chlorite in muscovite-quartz schists and results in its disappearance from the AFM diagram Fig 3(c).
- The diagnostic feature of this zone is the coexistence of cordierite and andalusite in common pelite compositions that plot below chlorite on the AFM diagram.
- All the chlorite had been consumed by reaction chlorite + muscovite -> cordierite + biotite + quartz + H2O before the conditions for reaction to form andalusite were attained. As a result, andalusite was produced at slightly higher temperature by the continuous reaction:
cordierite + muscovite + quartz-> biotite + andalusite + H2O
Sillimanite zone
- This zone can be ascribed to the occurrence of the polymorphic reaction:
andalusite →sillimanite.
Upper sillimanite zone
- An upper sillimanite zone with the assemblage sillimanite + cordierite + biotite + K-feldspar + quartz + muscovite is also present in this area, evidently resulting from reaction:
muscovite + quartz → Al2O3 + K-feldspar + H2O
Metamorphism of pelites at high pressures
- Recently a number of distinctive high-P, low-T assemblages have been identified.
- The question of the composition of pelitic rocks is particularly important in high-P metamorphism, because many high-P metamorphic belts are characterised by immature greywacke sediments and 'true pelites' of the type described in the earlier part of this chapter are very rare.
- Most of the high-P belts of the circum-Pacific region fall into this category.
- The best known characteristic of pelites in high-P terranes is the absence of biotite and the occurrence instead of phengite-rich muscovite.
- Garnet, chloritoid, kyanite and chlorite are also common Garnet may also be relatively rich in Mg. phengite and has been shown to be stable only at high pressures.
- A distinctive index mineral T, high-P metamorphism appears to be Mg-Fe-carpholite. Carpholite is otherwise known as a rare Mn mineral in medium pressure metamorphism.
- Mn-poor varieties have been found in pelites only from high-Pterranes. Carpholite can be considered as a lower grade equivalent of chloritoid, to which it may be converted on heating by the continuous reaction:
carpholite -> chloritoid + quartz + H2O
- Chopin & Schreyer (1983) have identified a sequence of four metamorphic zones in high-P rocks, and their assemblages are illustrated in Fig.4, projected from phengite + quartz + H2O.
- The lowest grade zone has pyrophyllite coexisting with chlorite and in some instances Fe-carpholite.
- At somewhat higher grades the association Fe-chloritoid + carpholite is found but pyrophyllite is still possible, although it may not coexist with chlorite, thereby distinguishing these rocks from lower pressure terranes.
- With further heating (Fig 4 (c), (d)) pyrophyllite is replaced by kyanite and chlorite progressively breaks down leading to the formation of distinctive Mg-rich chloritoid and garnet, and permitting the formation of talc in a wide range of pelitic rock types through reactions such as
Fe-chlorite + quartz -> garnet + talc + H2O
Fe-Mg-chlorite + quartz-> Fe-Mg-chloritoid + talc + H20
Mg-chlorite + quartz -> kyanite + talc + H20

13.3 Metamorphism of Calcareous Rocks
- Calcareous rocks are predominantly carbonate rocks, usually limestone (Ca carbonate) or dolostone (Ca-Mg carbonate).
- They typically form in a stable continental shelf environment along a passive margin.
- They may be pure carbonate, or they may contain variable amounts of other precipitates (e.g., chert or hematite) or detrital material (sand, clays, etc.).
- As the quantity of impurities increases, carbonates grade into calcareous clastic sediments (marls) and then into pelites or arenites.
- Carbonates typically become metamorphosed when the passive margin becomes part of an orogenic belt, either when it is transformed into an active margin by the development of a subduction zone inclined beneath the continent or when the margin enters a subduction complex as part of the subducting plate.
- Metacarbonates are metamorphosed calcareous rocks in which the carbonate component is predominant.
- Marbles are metamorphic rocks that are nearly pure carbonate.
- When carbonate is subordinate, the metamorphic rock may be composed of Ca-Mg-Fe-Al silicate minerals, such as diopside, grossular, Ca-amphiboles, vesuvianite, epidote, wollastonite, etc.
- Metamorphosed rocks composed of these minerals are called calc-silicate rocks.
- A skarn is a type of calc-silicate rock formed by metasomatic interactions between carbonates and silicate-rich rocks or fluids.
- This may occur at the contact between sedimentary layers but is developed most spectacularly at the contact between carbonate country rocks and a hot, hydrous, silicate intrusion, such as a granite.
- Metacarbonates compose only a small proportion of metamorphic rocks.
- Over the common range of medium and low P/T metamorphism, the only change in limestone marbles is an increase in grain size with grade (or in zones of fluid-enhanced recrystallization), and perhaps the development of a preferred orientation in some deformed rock bodies.
- They become more interesting only when metamorphosed at unusually high temperatures, when calcite breaks down to spurrite or or at high pressures, when it is transformed to aragonite.
- Metamorphism of impure limestones and dolostones, on the other hand, is far more interesting and informative.
- A number of Ca-Mg silicates can form in metamorphosed siliceous dolostones under commonly attainable metamorphic conditions.
- At the incipient stages of metamorphism, impure carbonates consist of a mixture of carbonate minerals (calcite, dolomite, and ankerite are most common), perhaps some chert, and detrital material such as fine quartz, feldspar, and hydrous minerals (usually clays).
- Metamorphism of these rocks is interesting because it involves several devolatilization reactions (typically both decarbonation and dehydration reactions) dependent not only on P, T, and the rock composition, but also on the composition of the associated pore fluid.
- To understand the metamorphism of these rocks we will have to adopt an approach that deals with these fluids as H2O-CO2
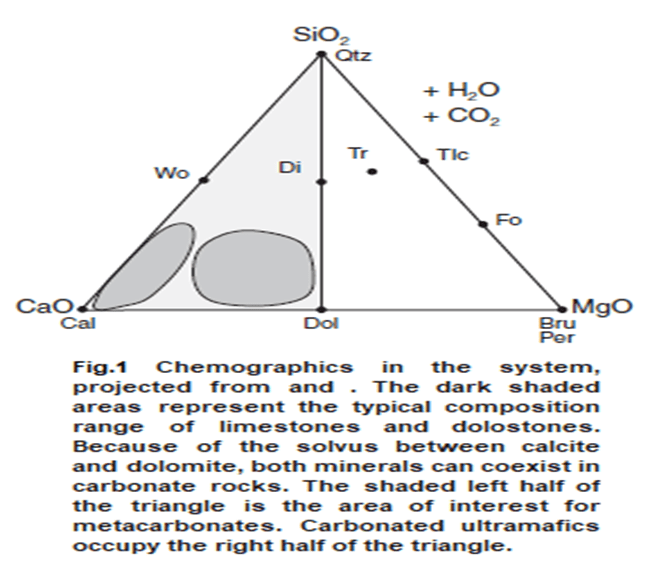
- We will concentrate on impure carbonates in which the major initial constituents are dolomite, calcite,and silica (chert, detrital quartz, or SiO2 in solution). Such rocks correspond nicely to the simple five-component system: CaO-MgO-SiO-H2O-CO2 (CMS-HC).
- When projected from the triangular CMS chemographic system depicts all the major minerals. Other components may also be present, typically FeO, Al2O3,K2O and Na2O but in most carbonate rocks they are present in only minor amounts and do not significantly alter the parageneses.
- Figure 1 illustrates the CMS system and the minerals that typically develop in metamorphosed siliceous carbonates.
- A comprehensive analysis of metamorphism of these rocks requires that we consider the reactions between these phases as a function of metamorphic grade on a petrogenetic grid, and the changes that the reactions have on the topology of CMS-HC compatibility diagrams.
- Because the composition of the fluid phase is so important in metacarbonates, adopt a T-XCO2 grid in favor of a T-P grid. To account for the effects of pressure, we can compare T-XCO2 diagrams at different pressures.
Contact Metamorphism of Siliceous Dolostones
- There are some differences between regional and contact metamorphism, the simplest sequence of zones that typically develops (in order of increasing grade) is:
- Talc zone
- Tremolite zone
- Diopside and/or forsterite zones
- To understand the development of these zones, we turn to the results of experiments in the CMS-HC system.
Marbles
Calcite Marbles
- The term marble is used for metamorphosed calcareous rocks in which carbonate minerals dominate.
- Many marbles are composed only of calcite with minor quartz and phyllosilicates, originally of detrital origin.
- There is sometimes graphite derived from organic debris, and pyrite is also a common accessory.
- The mineral assemblage in a marble of this type provides few clues as to the conditions of formation, since calcite is stable at all but the highest pressures, and even where aragonite does form during burial, it is likely in most cases to change back completely to calcite during uplift, except at very low temperatures.

- At very high temperatures and low pressures, calcite may react with any quartz present to produce calcium silicate, wollastonite.
- Despite the lack of mineralogical reaction in calcite marbles, they are susceptible to extensive textural changes due to recrystallisation of calcite to produce a coarser grain size and often a preferred orientation.
- The reaction to form wollastonite provides a simple example of one of the most common types of reaction to occur in carbonate rocks, i.e. a decarbonation reaction:
CaC03 + Si02--> CaSi03 + C02
- Like H20, C02 forms a supercritical fluid under metamorphic conditions, with a density that is broadly similar to that of supercritical water.
- Most wollastonite- occurrences are in thermal aureoles formed by contact metamorphism at relatively low pressures.
- Nevertheless, wollastonite is sometimes found in situations where it apparently formed at significantly higher pressures but without excessive temperatures.
- An explanation for these occurrences necessitates considering the possibility of metamorphic fluids intermediate in composition between H20 and C02.
Dolomitic Marbles
- The number of phases that can form from limestones composed only of CaC03 + quartz is limited.
- Only at exceptionally high temperatures and low pressures do other phases such as spurrite and lamite appear.
- Limestones that contain dolomite provide much more useful indicators of metamorphic grade because a range of Ca-Mg-silicates can form in the more usual P-T conditions of metamorphism, for example talc, tremolite and diopside.
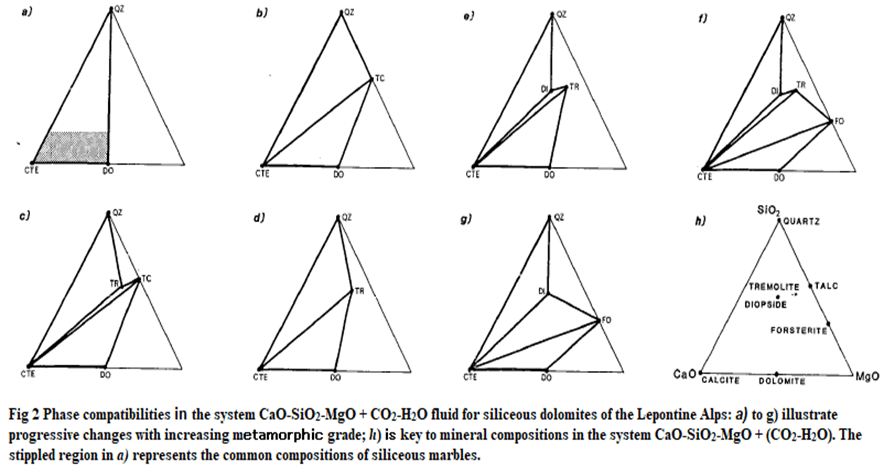
- The general sequence of mineral zonation in dolomitic marbles was first described by Eskola (1922) who first recognised the importance of talc at the lowest grades. The sequence of mineral-appearance isograds in regionally metamorphosed dolomitic limestones appears to be:
- - talc (not always present)
- -tremolite
- - diopside or forsterite
- - diopside + forsterite.
- Most earlier studies reported the appearance of forsterite before diopside, but the precise conditions for the growth of either mineral are dependent on rock composition, and since both appear at very similar temperatures, chance variation in lithology can dictate the relative order of appearance.
- For this reason, these two minerals have been grouped together here, although it is clear that higher temperatures are needed for them to coexist than for one or the other (according to rock composition) to occur.
- The mineral assemblages of impure dolomitic marbles can be conveniently represented in a triangular diagram with CaO, SiO2 and MgO at the apices.
- CO2 and H2O are treated as being available in excess to produce carbonate or hydrous phases.
- The locations of the common phases of metamorphosed marbles, plotted on such a diagram, are shown on Fig. 2(h).
- In addition to the Ca-Mg silicates and carbonates, and quartz, impure marbles can contain additional phases such as mica, feldspar, garnet, etc., which involve further components, but many marbles nevertheless have compositions that can be modelled very closely in the system Ca0-Mg0-Si02-C02-H20.
- Minor amounts of other phases do not substantially change the reactions among the Ca-Mg-silicates.
- The diagnostic three-phase assemblages found by Trommsdorf are represented graphically in Fig. 2, and from this diagram it is possible to suggest reactions to describe the changes between the zones, on the basis of the shift of the pattern of tie-lines.
- Figure 1(a) represents the original sedimentary assemblages of dolomite + calcite + quartz.
- The change between this diagram and Fig. 2(b), representing the low grade rocks is the replacement of the dolomite-quartz tie-line by a talc-calcite tie-line, and this change can be represented by the reaction:
3dolomite + 4quartz + 1H20→1talc + 3calcite + 3CO2
- Figure 2(c) represents the main paragenesis found when tremolite first reacts in. The composition of tremolite plots within the talc-calcite-quartz triangle of Fig. 2(b), from which we can deduce that the reaction is:
5talc + 6calcite + 4quartz -> 3tremolite + 2H20 + 6C02
- In the case of quartz-poor rocks, quartz may be completely consumed by this reaction to give the assemblage
talc + calcite + tremolite
- but in more siliceous rocks the talc is consumed to give
tremolite + calcite + quartz.
- Final disappearance of talc in these rocks can be ascribed to the reaction:
2talc + 3 calcite -> 1tremolite + 1dolomite + 1C02 + 1H20
- This gives rise to the phase relations illustrated in Fig. 2(d).
- At higher grades than the disappearance of talc + calcite, diopside + calcite or forsterite + calcite appear, although tremolite commonly persists.
- Figure 1(e) shows the appearance of the diopside within the tremolite-calcite-quartz field, through the reaction:
1tremolite + 3calcite + 2quartz-> 5diopside + 1H20 + 3CO2
- In contrast, the association of forsterite + calcite implies the replacement of the tremolite-dolomite tie-line (Fig. 1(f)) due to the reaction:
1 tremolite + 11 dolomite → 8 forsterite + 13 calcite + 1 H20 + 9 CO2
- It is clear from Fig. 1 that forsterite first appears in rocks whose compositions are silica-poor and lie near the base of the triangle, while diopside appears in relatively silica-rich or dolomite-poor rocks.
- Diopside and forsterite can coexist only when the tremolite-calcite tie-line has been removed, due to the reaction:
tremolite + 5 calcite → 11 diopside + 2 forsterite + 1 H20 + 5 C02
- This gives rise to the phase relations shown in Fig. 1(g).
- The natural sequence of assemblages, although conforming in a general way with the idealised sequence, is actually far more complex.
- Assemblages involving both reactants and products of these reactions may occur over large areas of countryside, and apparently low-grade assemblages may persist alongside higher grade assemblages Our simplified system has five components: CaO, MgO, Si02, H20 and C02.
- No assemblages have more than five phases, i.e. four solid phases plus a single mixed H20-C02 fluid phase.
- Hence even these assemblages have two degrees of freedom and can occur over a range of pressures and temperatures according to the composition of the fluid phase.
Controls on the Fluid Composition in Marbles
- We have seen in this chapter that the P-Tconditions at which many reactions in carbonate rocks take place are dependent on the composition of the fluid phase.
- However, the self-same reactions involve Components of the fluid and so change its composition.
- A marble with calcite + quartz may have a nearly pure H20 fluid phase present as it is heated until wollastonite begins to form, but at this point the fluid must become enriched in C02 from the decarbonation reaction.
- In the first case, no more water gains access to the marble after wollastonite starts to form.
- Further heating is accompanied by further increments of reaction which cause the fluid to become progressively enriched in C02 so that the fluid composition and temperature evolve together along the path of the isobaric univariant curve as long as calcite, quartz, wollastonite and fluid are all present in the rock.
- Fluid composition is controlled by the mineral assemblage of the rock itself and is described as being internally buffered.
- The second way in which fluid composition can be controlled in marbles is by external buffering.
- In this process, the fluid is derived from adjacent rock units or from magmatic processes, for example, and it is the source region that defines the fluid's composition.
- If such a fluid passes through the marble in sufficiently large quantities it may be able to flush away the fluid being generated by reactions going on within the marble without having its composition substantially modified. In this case the fluid composition is externally buffered.
- The reactions that take place in marbles and other carbonate-bearing rocks can be grouped into six types, each of which has a distinctive shape to the equilibrium curve on an isobaric T-X diagram.
- Decarbonation reactions
- Dehydration reactions
- Dehydration-decarbonation reactions Many reactions in marbles give off a mixture of C02 and H2O
- Hydration-decarbonation reactions: have H2O and C02 on different sides of the reaction
- Carbonation-dehydration reactions: the effect of increased temperature is to cause C02 to be consumed and H20 to be released. An example is the reaction: zoisite + COz ~ anorthite + calcite + H20
- Fluid -absent reactions Where no fluid is released or consumed.
Metamorphism of Calc-Silicate Rocks
- Calc-silicate rocks are rich in Ca, Mg, Fe, Si, and Al. They are generally more common in metamorphic terranes than siliceous limestone and dolostone marbles. Many calc-silicate rocks begin as carbonate-bearing pelitic sediments (sometimes called calcareous pelites, or marls).
- The clastic component may include quartz, feldspar, clays, and other silicates or oxides. The proportions of clastic and carbonate minerals can vary widely.
- When carbonate is subordinate to other clastic material, it may be largely consumed by decarbonation reactions, and the metamorphic rocks are then composed solely of Ca-Mg-Fe-Al-Na-K silicate minerals (calc-silicate minerals), such as diopside, the epidote group minerals, grossular, Ca amphiboles, calcic plagioclase. Muscovite, biotite, and chlorite are also common.
- Some calc-silicate minerals (e.g., zoisite, margarite, vesuvianite, and grossular) are stable in whereas others (e.g., anorthite) are stable in CO2-rich fluids.
- The range of calc-silicate bulk rock compositions overlaps with that of mafic to intermediate volcanic (and volcaniclastics), so that the metamorphic rocks may not always be distinguishable petrographically.
- Five zones in the calcsilicate rocks are described.
- The reactions that define the isograds and the continuous reactions within the zones are complex, and several equilibria may occur simultaneously.
- The reactions listed below are only approximations, intended as simplified representations.
- ANKERITE ZONE Rocks of the lowest grade, the area, contain the assemblage ankerite quartz albite muscovite calcite chlorite. Ankeritic carbonate is a dolomite with over replacing Mg and is stable in sedimentary environments.
- BIOTITE ZONE The biotite isograd is related to the inferred reaction:
Ms+Qtz+Ank+H2O = Cal+Chl+Bt+CO2
- It occurs at about the same grade as the biotite isograd in the local pelites.
- Calcareous rocks in the lower biotite zone have the mineral assemblage
biotite + ankerite + quartz + albite + muscovite + calcite + chlorite
- AMPHIBOLE ZONE Ca-amphibole is inferred to be generated by the reaction:
Chl + Cal + Qtz + Ab = Act + An + H2O
- A typical mineral assemblage in the zone is Ca-amphibole + quartz + plagioclase + calcite + biotie ± chlorite.
- The amphibole is tremolite-actinolite in the lower zone, but becomes more aluminous across the zone. The average plagioclase composition in the zone is .
- ZOISITE ZONE Zoisite (a nearly Fe-free epidote mineral) first appears as reaction rims on plagioclase at its contact with calcite, suggesting the reaction:
An + Cal + H2O = Zo + CO2
- The typical mineral assemblage is zoisite Ca amphibole + quartz + plagioclase (average An74) + calcite ± microcline.
- DIOPSIDE ZONE The diopside isograd in the calcareous rocks occurs at about the grade of the sillimanite isograd in the pelites. Diopside growth results from dehydration of amphibole at high grade caused by the reaction:
Tr + Cal + Qtz = Di + H2O + CO2
- The typical mineral assemblage is diopside + zoisite Ca-amphibole + calcite + quartz + plagioclase (avg. An79) ± biotite ± microcline.
- Ca-amphibole continues to coexist with diopside in the zone because of the continuous nature of reaction Tr + 2 Qtz + 3 Cal = 5 Di + 3 CO2 + H2O.

- The five zones observed by Ferry are common, but not universal, in calc-silicate rocks. Differences in P, T, bulk composition, and fluid composition may result in the development of several other mineral zones. steep.
- Infiltration, and the chemical changes that accompany it, may be widespread in metamorphic rocks, especially at the higher grades. It has been documented in many calcsilicate rocks, where the effects are most easily recognized.
- Fluids play an influential role in many igneous and metamorphic processes, where they may partake in various types of reactions or act as a transporting medium for the movement of soluble material. processes.
- Fluids are essential components in pegmatite and ore genesis as well. Fluids can change the transport properties of a rock by dissolution or precipitation of material, or by creating fractures due to fluid pressure (“hydrofracturing”).
- Fluids can also affect the mechanical properties of rocks, and therefore the nature and extent of deformation in areas under stress.
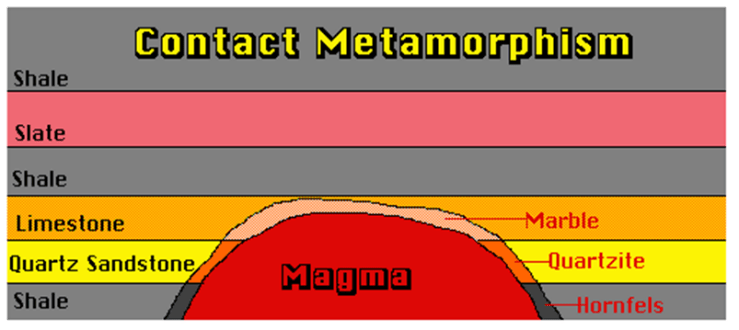
13.4 Metamorphism of Sandstone
- Quartzite is a non foliated metamorphic rock composed almost entirely of quartz.
- It forms when a quartz-rich sandstone is altered by the heat, pressure, and chemical activity of metamorphism.
- Metamorphism recrystallizes the sand grains and the silica cement that binds them together. The result is a network of interlocking quartz grains of incredible strength.
- Most quartzite forms during mountain-building events at convergent plate boundaries where sandstone was deposited on a continental plate.
- There, the sandstone is metamorphosed into quartzite by the intense pressure of a plate collision and often by deep burial.
- Compressional forces at the plate boundary fold and fault the rocks and thicken the crust into a mountain range.
- Quartzite is an important rock type in folded mountain ranges throughout the world.
- They are also formed due to contact metamorphism.