Generation and Crystallization of Magmas
Magma
Introduction
- Most magmas originate by melting in the mantle, but some show evidence of partial crustal component.
- Magma is the underground molten rock. When the magma comes out to earth surface, it is known as Lava.
- The generation of magma is linked up with the global tectonics. Most of the active volcanoes are associated with divergent and convergent plate boundaries. Example of active volcano: Hawaii Island.
- The partial melting of silicates rocks of earth’s mantle and crust (both continental+ oceanic) is responsible for the formation of magma.
- Magma composition can be either felsic or basic depending upon the origin in earth’s interior.
- Ocean crust= Basaltic magma, Continent crust= Rhyolitic-andesitic magma.
- Most of the magma comes from the mantle by melting of peridotite.
- Magmas do not come from the only liquid part of the Earth, the outer core, because it does not have the right chemical composition. The outer core is made mostly of Fe with some Ni, magmas are silicate liquids.
Petrology of mantle
- The mantle is composed mostly of mafic silicate minerals aka Peridotites (olivine + pyroxene).
- Peridotite is a generic name used for coarse-grained, ultramafic igneous rocks which contains olivine as its primary mineral, along with other mafic minerals such as pyroxenes and amphiboles.
- The peridotite "family" contains several different intrusive igneous rocks. These include lherzolite, harzburgite, dunite, wehrlite, and kimberlite.
- Only direct samples can provide us with an accurate idea of mantle composition.
Surface samples generally accepted to be of mantle origin come from the following sources:
a. Ophiolites: these are large sheet-like mafic to ultramafic masses, presumed to be of ancient oceanic crust and upper mantle thrust onto the edge of the continents and/or incorporated into mountain belts.
- The original mineralogy is dominated by olivine, orthopyroxene, clinopyroxene and lesser amounts of plagioclase and oxide minerals including magnetite, ilmenite, and chrome-rich spinel.
b. Dredge samples from oceanic fracture zones: Slow-spreading ridges have extensive detachment zones exposing deeper mantle.
- Samples collected along these zones only represent the uppermost mantle.
- Samples collected are of same type as ophiolites, thus confirming the evidence that ophiolites are samples of oceanic crust and upper mantle.
c. Nodules in basalts: Ultramafic xenoliths, called nodules, are occasionally carried to the surface by basalts.
d. Xenoliths in kimberlite bodies: Kimberlites tap an upper mantle source as deep as 250 to 350 km and travel rapidly to the surface, bringing a variety of mantle and crustal samples to the surface as xenoliths. Thus, they are the only source of samples below the uppermost layers.
- To gain insight into mantle petrology, we choose some mantle material and subject it to high pressure and temperature experimentation.
- The best-known example of a synthetic mantle analogue is Pyrolite, a pyroxene-olivine material.
Types of magma
Primary Magmas
- These are formed at depth and not modify subsequently which means no differentiation take place here.
- Primary melt obtained when the rock melts.
- They are also called primitive type of magma.
- Example: Leucosomes of migmatites
Parental Magma
- This type of magma forms the base from where the observed range of magma can be derived by the process of igneous differentiation.
- It is not of primitive type.
- Example: At high degree of partial melting generally komatite and picrite is produced.
Derivative magmas are of modified type and generally evolved from parental magma.
Physical properties of magma
Magmatic temperature: Since a rock is a mixture of minerals, it does not have a single melting temperature.
- It melts over a temperature range that is commonly several hundred degrees.
- Most magma is a mixture of crystals and liquid. If the proportion of crystals is small, we describe the magma as having the properties of a suspension, which are predominantly those of a liquid.
- If the proportion of crystals is large enough that the crystals we describe the magma as a mush, which has properties closer to those of a solid than to a liquid yet is still capable of moving.
- All magmas pass through the mush stage during cooling.
- Increased pressure causes melting temperatures to rise, but if water is present, the increased pressure allows water to dissolve in the magma, which in tum significantly lowers the melting temperatures.
- Once the magma reaches the surface, it starts to cool by radiation (forms crust immediately) and conduction (cooling withing the magma is slow).
- Several factors determine the cooling rate of magma, the most important of which are thermal gradients, rate of heat diffusion through magma and rock, heat capacity of magma and surrounding rocks, and the shape of magmatic bodies.
Magma Densities: density affects a number of petrologic processes like buoyancy, differentiation, magma mixing.
Magma Viscosity: Viscosity is the physical property that describes this resistance to flow. Viscosity is extremely important in determining the rates of emplacement of 1nagn1a and the shapes of igneous bodies, and in detern1ining whether sinking or floating crystals will separate fast enough from their parent magma to change the magma's bulk composition.
- Common magmas cover a wide range of viscosities. At a temperature of 1200 °C and atmospheric pressure, basaltic magmas (gabbroic) have viscosities in the range 10 to 100 Pa s.
- Most rhyolitic magmas (granitic) have viscosities above 105 Pa s and can be as high as 108 Pas at 800 °C.
- The viscosity of a magma is determined largely by its chemical composition, which controls its degree of polymerization.
- Just as silicate minerals show different degrees of polymerization, so do silicate melts.
- The increase in viscosity from komatiite through basalt and andesite to rhyolite is attributable to this chemical variation, with komatiites containing approximately 40-45 wt% Si02, basalts -50 wt% Si02, andesites -60 wt%, and rhyolites -73 wt%.
- Temperature also affects viscosity. Lower temperature, more viscosity.
- Rhyolitic magma is typically several hundred degrees cooler than basaltic magma and consequently has much higher viscosity than basalt.
- This fact is the reason for morphological differences observed in different structures formed from different magmas. Eg. Volcano shapes.
- Pressure has little effect directly over viscosity but it, modifies viscosity indirectly.
- Higher pressure permits more water to be dissolved in magmas, which depolymerizes the melt and lowers the viscosity.
Generation of Magma
Methods of Generation of Magma
There are 3 basic ways to melt the mantle to generate molten magma:
- Heat-induced melting caused by increasing the temperature.
- Decompression melting caused by lowering the pressure,
- Flux melting caused by adding volatiles
Heat induced melting (Raising the temperature)
- It is the process of heating the mantle above the normal geotherm. This is achieved by accumulating enough heat by the decay of radioactive elements.
- The melting generated by heat induced thus transform solid mantle into liquid magma by simply applying heat.
- Heat-induced melting occurs at the mantle plumes or hotspots.
- Migmatites are also the result of heat induced melting generally formed by partial molten metamorphic rock.
- The magma that forms this rock shows the character of hybrid metamorphic-igneous type.
Migmatite
Decompression melting (Lowering pressure)
- A feasible way to lower the pressure is to raise the mantle rocks to shallower levels while maintaining their stored heat content.
- At the MORs, the magmas are produced by decompression melting.
- The rapid upliftment would minimize the heat loss, eventually intersecting the solidus and initiating melting.
- The convection currents generate in the asthenosphere is responsible for this.
- The lithosphere’s upper portion (crust) is a poor heat conductor, so the temperature remains constant throughout the underlying mantle material.
- The convection currents cause mantle material to rise, the pressure decreases, which cause the melting point to drop.
Convection in mantle
Flux melting
- It is achieved by adding volatiles or changing the composition.
- The volatiles are mobile and can have an effect on melting of the mantle.
- We find fluid inclusions in mantle minerals which are composed mainly of H2O and CO2.
- The melting take place when water and carbon dioxide is mixed in rock, thus helping the rock to melt at low temperature.
- Many of the volcanoes in the circum-Pacific subduction zones (Ring of Fire) are produced by flux-melted magma.
- The flux melting is generally take place in subduction zones where island arcs where volatiles are added to the liquid magma.
Flux melting at subduction zone
Origin of Magma
- The variation of temperature and pressure can be seen with respect to depth inside the earth.
- The heat inside the earth is due to the residual heat left due to the earth’s formation and due to the radioactive decay.
- GEOTHERMAL GRADIENT: The rate at which temperature increases with depth.
- The average geothermal gradient in the upper 100 km (62 mi) of the crust is about 25°C per km of depth.
- With increase in every kilometre in depth the temperature also ascends by 25°C.
- Beneath the oceans the normal geothermal is higher as compare to beneath the continents at shallow depths.

Geothermal gradient
- Partial melting is mostly applicable as it requires less in increment in temperature and less of decrease of peridotite
- Solidus= The temperature at which any crystal is solid and melting begins.
- Liquidus= The temperature where the complete melting of crystal take place.

Solidus line graph
- Due to the mobile nature of liquid it can be separated from the remaining solids once the partial melting has formed. The liquids generally displayed low density as compare to solids.
- The melting temperature of any substance is depends upon pressure so the solidus line slope is towards the right in the graph.
- The higher pressure created at depth thus increase the temperature which helps in melting of rock.
Origin of Basaltic Magma
- Mid-oceanic ridges are the regions where the basaltic magma generates i.e. mid-ocean ridge basalts (MORBs) as well as in scattered intraplate volcanic centers creating oceanic islands.
- Alkaline basalts are more found in intraplate centres while tholeittic basalt are found in submarine volcanism thus helps in formation of oceanic crust and are more voluminous.
- Alkali basalt: They are silica deficient and formed by low degree of partial melting and consist of CO2 rich volatiles at deep inside the earth. They are formed by deep-seated fractionation of Al-rich silicate phases.
- Theoleittic basalt: These are formed by shallow melting or by olivine fractionation and generally consist of water rich volatiles.
- Both magmas are evolved to give silicate type of magma in distinct pathways.
- Re-melting of the crust is always being there before it reaches the surface of the earth.
- The temperature at which these basalt magmas are found is between 1100°C to 1200°C.
- Subsequent crystal fractionation occurs during the post-segregation ascent can helps in the composition of basaltic magma.
Magmatic processes
- A range of basaltic magmas could be created by partially melting the mantle.
- Differentiation is an important natural process, capable of modifying the composition of magma and leading to a spectrum of igneous rocks.
- Diversification involves separating different phases of contrasting composition.
- This separation can happen during melting or during crystallization, when phases in different states coexist.
Partial melting
- Separation of a liquid from the partially melted solid residue is a form of diversification because it involves partitioning and separation of chemical constituents, and it can produce a variety of melt compositions from a single source.
- The major element composition of this melt remains constant as more is produced, until one of the source mineral phases is consumed by the melting process.
- Once a phase is consumed, the next melt increment will occur at a different composition and temperature because it now corresponds to the minimum melt in a reduced system with one fewer component and phase.
- Different melts can be extracted from an initially uniform source rock.
- When a rock begins to melt, a tiny fraction of initial melt forms discrete liquid drops at the junctions of mineral grains, usually at the points where three or four grains meet.
- Only when a critical quantity of melt is produced will there be a sufficient liquid volume that:
- The liquid forms an interconnected network.
- The interior body of the liquid can be free from the restraining effects of crystal surface adsorption.
- Only when conditions 1 and 2 are met can some of the melt be separable from the solids.
- The critical melt fraction required to form an interconnected network depends upon the dihedral angle, , formed between two solid grains and the melt.
- If < 60 , the melt may form a network with as little as~1% melt.
- As the dihedral angle increases above 60°, the amount of melt required to establish connectivity also increases.
- Viscosity is an important factor in melt segregation, once a continuous network has been formed.
- High-viscosity silicic melts, such as granitic–rhyolitic liquids, are less easily extracted.
- The critical melt fraction, or rheological critical melt percentage (RCMP), is the percentage of melt at which a crystal-dominated, more rigid granular framework gives way to a melt-dominated, fluid suspension, commonly called a crystal mush.
- Liquid separation is commonly motivated by gravitational effects as the buoyant liquid seeks to rise and escape the crystal residue.
- Because melt source regions are generally deep and under pressure, the separation is aided by a process known as filter pressing, or compaction.
- Here the crystal–liquid system is squeezed like a sponge, and the liquid migrates from the compacted solids.
Igneous–tectonic association
- The term refers to broad types of igneous occurrence, such as mid-ocean ridge, island arc, and intra-continental alkalic systems.
- Plate tectonics plays a major role in the generation of several magma types. Other types result from processes at depths.
- Magma types and their geologic setting is shown in figure.
- Most voluminous activity occurs at divergent plate boundaries (MORs).
- Shallow mantle beneath the ridge undergoes partial melting, resulting basaltic magma rises above and forms the oceanic crust (location 1).
- If the divergent boundary is present is beneath the continental crust (location 2), alkaline magmas are produced initially.
- As rifting continues, oceanic crust forms between the separating continental blocks.
- An oceanic plate created at mid-ocean ridges moves laterally and eventually is subducted beneath a continental or another oceanic plate.
- Melting also takes place at these subduction zones and the number of possible sources of magma here is more than at ridges.
- Sources may include various components of mantle, subducted crust, or subducted sediments.
- Thus, the types of magma produced are more variable than for divergent boundaries, but andesites are the most common.
- If oceanic crust is subducted beneath oceanic crust (location 3), a volcanic island arc forms.
- If oceanic crust is subducted beneath a continental edge (location 4), a continental arc forms along the active continental margin.
- Mineralogically continental arc is more silicic than oceanic arc.
- Location 5 is the site for back-arc magmatism.
- Back-arc occurs due to fractional drag associated with subducting plate.
- Such drag pulls down part of the overlying mantle, requiring replenishment from behind and below.
- Back-arc magmatism is similar to MORs but the spreading is slower, volcanism is irregular and less voluminous leading to thinner crust.
- Some igneous activity occurs within the plates, both oceanic (location 6) and continental (location 7).
- Source of the melt is deep probably in the asthenosphere. Hence, plate tectonics has no role to play here.
- Ocean islands such as Hawaii, the Galapagos, and the Azores all form via volcanism within the oceanic plates.
- Sometimes, the igneous activity gets progressively younger in one direction.
- There is a correlation between the direction and plate motion, indicating that the plate is moving over a hotspot (rising plumes that can penetrate either oceanic or continental lithospheric plates).
- Thus, we observe that the magmas reaching the surface are highly varied, ranging in composition from alkaline to acidic.
- Explaining the origin of this diversity is the dominant goal of petrology.
- L. Brown argued that basalt was the primary magma from which other magmas were derived.
- Igneous rocks related to a common source are said to be comagmatic.
Crystallization of magmas
Introduction
Crystallization is a process whereby solid crystals are formed from another phase, typically a liquid solution or melt. The crystallization sequence is given by Bowen’s reaction series. Kindly discuss this series, when you are writing about the crystallization.
General observation from experimental data shows us that:
- Cooling melts crystallize from a liquid to a solid over a range of temperature.
- Several mineral phases crystallize over temperature range, and the number of minerals tends to increase as temperature decreases.
- Minerals usually crystallize sequentially, generally with considerable overlap.
- Minerals involve solid solution change composition as cooling progresses.
- The melt composition also changes during crystallization.
- The minerals that crystallize, as well as the sequence in which they form, depend on the temperature and composition of the melt.
- Pressure can affect the temperature range at which a melt crystallizes. It may also affect the minerals that crystallize.
- The nature and pressure of any volatile components (such as H2O or CO2) can also affect the temperature range of crystallization and the mineral sequence.
Magam melts and crystal formation
- What is mineral?
- Naturally occurring substance.
- Consist of definite crystal structure and chemical composition.
- Occurs in rocks inside the earth crust.
- Rock textures are formed due to the aggregates of minerals which tell us about their trends of magmatic evolution.
- Igneous rocks crystallize from molten rock called magma.
- Magmas consist of hot silicate / carbonate /oxide/ phosphate/ sulphide and sulphur melts from which igneous rocks form.
- Partial melting of deep-seated rocks at depth of earth is responsible for formation of different types of magma.
- Primary melts
- Forms when the rock melts so that they form liquid melt.
- They are composed of suspended crystals and dissolved gases.
- Major silicate oxides exist in magma are SiO2, Al2O3, Fe2O3, FeO, MgO, CaO, Na2O and K2O, as well as from lesser proportions of TiO2, P2O5, H2O, CO2, S ,trace elements and other.
- Volatiles and the silicate components which are soluble are concentrated in the remaining liquid as the crystallization proceeds thus helping in the fluidity of the magma.
- Those minerals which are crystallized earlier from the magma are denser than the magma and due to buoyancy the gravity settling helps in setting of these crystals.
- As there is decrease in temperature take place the viscosity of silicate magma increases with decreases in dissolved H2O thus increasing the crystal fraction and an increase in the degree of SiO2 content.
Crystal formation of rock forming minerals
- All igneous and metamorphic rocks are composed of crystalline mineral grains.
- Crystal growth: The well developed crystals forms from time to time when the magma cools down.
- Grain size of the crystal affects the rheology of rocks and thus plays important roles in determining flow rates in the mantle which affect many other geological processes through plate tectonics.
- Until there is the existence of further cooling to a supersaturation level, the crystallization of mineral do not occur.
- The process of nucleation began when a stable nuclei of crystal tends to form and grow further.
- The temperature at which the crystal nucleate is less than at which they would melt in the magma so the liquid is at its meta-stable form and thus resemble the supercooled stage.
- The supersaturation acts like a driving mechanism for the growth of a crystal. As the degree of supersaturation increases the growth rate becomes faster.
- Material that is too far from the nucleus generally forms the separate nucleus.
Factors affecting crystallization mechanism
- The initial bulk composition of the melt is responsible for determining the composition paths followed by both the melts and the growing crystal depend.
- The composition of both the melt and the crystal also dependent upon at what level the crystallization process has been completed.
- There is continuity seen in the crystallization of minerals in solid solution series with the composition of the mineral changing along with that of the magmatic liquid.
- There is discontinuity seen in the crystallization of minerals which do not show solid solution series.
- As the melt reaches the appropriate temperature the discrete minerals will appear suddenly.
- Minerals that crystallize at high temperatures are rich in Mg and Fe and relatively poor in Si.
- Minerals that crystallize at the lowest temperatures are rich in Si and Al.
- Intermediate minerals that crystallize at intermediate temperatures are intermediate in relation to Si.
- Minerals crystallize at higher temperature exhibit lees polymerization of silica-tetrahedron as compare to lower temperature minerals.
- Degree of polymerization increases from orthosilicates to tectosilicates.
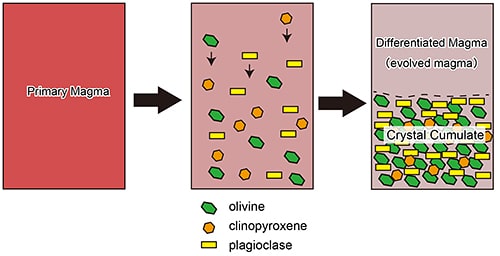
Formation of differentiation magma
Crystal Growth Rates
- As the nucleus becomes stable and continuous to grow in size thus forms the crystal whose size is determined by the nutrients concentration present in their surrounding and the proximity of the nuclei of neighbors.
- Four steps are involved in the growth process-
- The diffusion of nutrient to the nucleus through the medium takes place when the crystal is tending towards growth thus the rate of diffusion vary w.r.t the medium such as gas, liquid, silica melt or solid rock.
- Once the nutrients arrive at the nucleus the reaction take place and arrange themselves into building units that are acceptable to the crystal.
- The building units must then attach themselves to the crystal surface which helps in nucleation of the new surfaces or the growth of dislocations.
- The heat of crystallization is finally produced by thse builing units by attachment and there increase in components concentration do not enter the crystal thus both will dissipate before the further growth rate can occur.