Magmatic Differentation and Assimilation
Magmatic Differentation and Assimilation
Magmatic Differentiation
PYQs: Magmatic differentiation and assimilation
- What is magmatic differentiation? Discuss various differentiation processes involved in magma modification. (IFS 2021, 15)
- Discuss briefly the processes of magma generation in the Earth's interior. How is grain size of an igneous rock related to the rate of cooling of magma? Discuss the role of fractional crystallization and assimilation in magmatic differentiation. (2021/20 marks)
- Differentiate between magmatic differentiation and assimilation. 10-2016
- Explain magmatic differentiation during crystallization of magma and its petrogenetic significance. 8-2014
- Write in 150 words: magmatic differentiation. 10-2007
- Describe the processes of magmatic crystallization and assimilation. Illustrate your answer with examples from ultrabasic rocks and alkaline rocks. 40-2003
- Write explanatory notes: Bowen’s Reaction Principle and magmatic differentiation process. 30-2001
Introduction
- Magmatic differentiation is process through which a single homogeneous magma produces two or more magma fractions of different composition (daughter magmas), which ultimately forms diverse rock types.
- It is the process by which magma is able to diversify and produce a magma or rock of different composition.
Why magma differentiated?
- Diversification of magma involves the separation of different phases of contrasting composition of magma.
- This separation can take place during melting or during crystallization, when phases in different states coexist.
- The requirements for the diversification of magma in geologic systems-
- Presence of two or more phases in which components are distributed unequally.
- Phases are separated in the physical process.
- The chemical reaction occurs between the magma and the crystals which are first to solidify from it. It is responsible for the differentiation of magma.
- The differentiation also occurs due to the physical separation of the first crystals form during the remaining magma.
- The major causes of magma differentiation are- Cooling and Contamination.
- Cooling: The cooling causes the different minerals to crystallize from the melt of magma, as determined by the Bowen principle.
- Contamination: It is represented in assimilation. It is caused by the wall rock assimilation i.e. the mixing of different magmas in the magma chamber.
The process
- Differentiation of magma is responsible to produce the magma series sequentially. It can be explained by Bowen series.
- Partial melting is the melting of the portion of solid in the magma chamber. This melting is responsible for the different solidus and liquidus temperature.
- Differentiation (and partial melting) involves two essential processes:
- Compositional differences are created between the phases as the various elements are broken themselves due to change in intensive factors. Eg. pressure, temperature, or composition. This determines the trend of the differentiation process.
- Preservation of the chemical difference created in part 1 by segregating the chemically distinct portions, which then evolve as separate systems.
- Fractionation is the physical process by which different portions or phases are mechanically separated. Its effectiveness determines the extent to which differentiation proceeds along a particular trend.
- The most common forms of magmatic differentiation involve the physical separation of phases in multiphase systems.
- The effectiveness of this separation depends upon contrasts between the phases in physical properties such as density, viscosity, and size/shape.
- The energy providing the force for the separation is usually thermal or gravitational.
- The fractionated phases in magmatic systems can be either liquid–solid, liquid–liquid, or liquid–vapor.
Fractional Crystallization
- With crystallization we see change in liquid composition (except in eutectic composition). If the crystals are removed, then different magma compositions can be generated from the initial parent liquid.
- Thus, fractional crystallization can produce liquid compositions that would not otherwise have been attained by normal crystallization of the parent liquid.
- A very good example of this process is Bowen’s reaction series.
Mechanism of fractional crystallization
- For the process to operate, crystals have to be removed from the magma so that they can no longer interact with the liquid.
- Gravity settling/crystal settling: crystals formed have different densities, thus, high density crystals will sink to the bottom of the magma chamber. with progressive crystallization, earlier formed layers will get removed from the liquid.
- cumulate texture is a result of fractional crystallization.
- Filter pressing (compaction): is also possible in crystal mushes that form as cumulates or crystal suspensions.
- With the added weight of further accumulation, the crystal mush may be compacted squeezing much of the liquid out into the main magma body.
- Another method of filter pressing involves the movement of a phenocryst-laden crystal mush. Any constriction in the conduit causes the crystals to interfere and slow with respect to the liquid.
- Another mechanism by which crystals may be segregated from the liquid occurs when crystal-rich magmas flow in a laminar fashion near the walls of the magma body. The process is known as flow segregation.
- The motion of the magma past the stationary walls of country rock creates shear in the viscous liquid as a result of the velocity gradient near the walls.
- The resulting differential motion forces the magma to flow around phenocrysts, thereby exerting pressure on them at constrictions where phenocrysts are near one another or near the contact itself.
- The pressure, called grain dispersive pressure, forces the grains apart and away from the contact.
- This effect is greatest near the walls, and it drops off quickly toward the magma interior.
- Phenocrysts thus concentrate away from the walls to mitigate the pressure buildup. This concentration is most apparent in dikes and sills.
- Inward Crystallization – As magma body is hot and the country rock which surrounds it is cooler, heat will move away from magma.
- thus, crystallization is expected to take place in this cooler portion near the walls. the magma would then be expected to crystallize from the walls inward.
Volatile Transport
- Chemical differentiation can also be accomplished when a separate vapor phase coexists with a magma and liquid–vapor fractionation takes place.
- A vapor phase may be introduced in any of three principal ways.
- First, a fluid may be released by heating of hydrated or carbonated wall rocks.
- Second, as a volatile-bearing but undersaturated magma rises and pressure is reduced, the magma may eventually become saturated in the vapor, and a free vapor phase is released. Because the vapor phase has a lower density than the melt, it rises, diffusing through the magma, and concentrates near the top of the magma chamber. Such concentrated fluid may even permeate into the roof rocks. This process usually involves an H2O-rich fluid, and it produces a variety of hydrothermal alteration effects.
- third mechanism for generating a separate fluid
- phase is a result of late-stage fractional crystallization. Most early formed igneous minerals are anhydrous, so their segregation from a hydrous melt enriches the melt in H2O and other volatile phases. Eventually the magma reaches the saturation point, and a hydrous vapor phase is produced.
Liquid Immiscibility
- Liquid immiscibility is where liquids do not mix with each other. Eg. water and oil.
- Immiscibility is temperature dependent.
- If immiscibility is to produce sizable bodies of contrasting magmas, it appears necessary for magma first to crystallize fractionally, so that when unmixing does take place, the conjugate liquids are free to separate.
- Three natural magmatic systems are widely recognized as having immiscible liquids in some portion of their compositional range.
- The first is the natural Fe-rich tholeiitic basalts, which experience an initial trend toward iron enrichment.
- In the later stages of fractionation, a “granitic” melt (>75% SiO2) separates from a basaltic melt ( 40% SiO2 ).
- Once separated, the silicic liquid must have a much lower density than the Fe-rich mafic liquid, and we would expect it to rise and collect near the top of the magma chamber.
- A second system displaying immiscible liquid behavior is the separation of a sulfide-rich liquid from a sulfide-saturated silicate magma. The solubility of sulfides in silicate melts is extremely small.
- A third liquid immiscibility gap occurs in highly alkaline magmas that are rich in CO2. These liquids separate into two fractions, one enriched in silica and alkalis and the other in carbonate.
Magma Mixing
- If two or more magmas with different chemical compositions come in contact with one another beneath the surface of the Earth, then it is possible that they could mix with each other to produce compositions intermediate between the end members.
- The commingling of magmas may result in a range of products.
- Magmas may not mix at all but simply remain as discrete bodies or xenoliths of one in the other.
- At the other extreme, they may mix completely to form a homogeneous hybrid magma. The bulk density of this hybrid magma is intermediate between the other two magmas, and it therefore accumulates between them.
- The dynamics of magma mixing depend on the contrasting magma properties, such as the temperature, composition, density, volatile content, and viscosity, as well as the location and the turbulence with which one magma injects into the chamber containing the other.
- Density contrast would lead to the lighter one floating over the denser one.
- Temperature contrast would lead to cooling of the one with lower temperature.
- Viscosity contrast would necessitate some kind of vigorous stirring or a turbulent plume for the magmas to mix. This is provided by convection.
- The smaller the difference in chemical composition between two magmas, the smaller will be the contrasts in temperature, density, and viscosity.
- Some magma mixing occurrences may appear like immiscible liquids, but the mixed magmas are not in equilibrium, so disequilibrium assemblages are common.
- Mixing of mantle and crustal melts may be an important process in the evolution of many subduction- related calc-alkaline magmas.
Evidence for Mixing
- Mingling of magmas: dark coloured mafic rock mingling with light-coloured rhyolitic rocks.
- Disequilibrium Mineral Assemblages: if a basaltic magma containing Mg-rich olivine mixed with a rhyolite magma containing quartz, and the magma was erupted before the quartz or olivine could be redissolved or made over into another mineral, then we would produce a rock containing mineral that are out of equilibrium.
- Reverse Zoning in Minerals: Mg-Fe solid solution minerals normally zone from Mg-rich cores to Fe-rich rims. If a Fe-rich olivine or pyroxene is mixed into a Mg-rich magma that is precipitating Mg-rich olivine or pyroxene, it may precipitate the more Mg-rich composition on the rims of the added crystals.
- Glass Inclusions: Sometimes the crystal grows incompletely trapping liquid inside. If that liquid is quenched on the surface and a thin section is cut through the crystal this trapped liquid will be revealed as glass inclusions in the crystal. If the composition of glass inclusions is different from glass in the groundmass, and if the groundmass composition is not what is expected from normal crystallization of the minerals present, this provides evidence of magma mixing.
Magma Assimilation
PYQs: Magmatic differentiation and assimilation
- What is magmatic differentiation? Discuss various differentiation processes involved in magma modification. (IFS 2021, 15)
- Discuss briefly the processes of magma generation in the Earth's interior. How is grain size of an igneous rock related to the rate of cooling of magma? Discuss the role of fractional crystallization and assimilation in magmatic differentiation. (2021/20 marks)
- Differentiate between magmatic differentiation and assimilation. 10-2016
- Explain magmatic differentiation during crystallization of magma and its petrogenetic significance. 8-2014
- Write in 150 words: magmatic differentiation. 10-2007
- Describe the processes of magmatic crystallization and assimilation. Illustrate your answer with examples from ultrabasic rocks and alkaline rocks. 40-2003
- Write explanatory notes: Bowen’s Reaction Principle and magmatic differentiation process. 30-2001
Introduction
- Assimilation is the process whereby foreign material is incorporated into magma.
- It is the process of reacting of the magma with the wall rock, whereby the country rock are incorporated in the magma and eventually melt.
- It can extensively alter the composition of magma.
- Evidence of contamination of magma through the assimilation of country rocks can be found at the margins of many
- Evidence of contamination of magma through the assimilation of country rocks can be found at the margins of many Eg. xenoliths (foreign materials) suspended in igneous rocks.
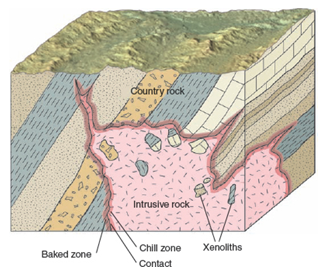
- When we observe minerals are produced that could not possibly form if there had been simply fractional crystallization. Quartz, for example, can be found near the margins of nepheline-bearing intrusions that were contaminated by quartz-bearing sedimentary rocks.
The process of assimilation
- Assimilation consists of 3 processes-
- Melting of country rock
- Dissolution/diffusion of country rock
- Reaction between magma fluid and country rock
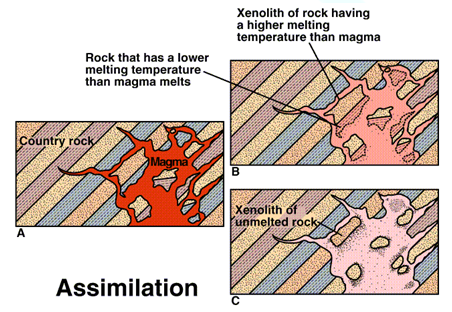
- The degree to which a magma can assimilate the country rock by melting is limited by the heat available in the magma.
- The country rock must be heated to the melting point first and then at least partially melted in order to be assimilated.
- The heat required (to raise the temperature and supply the latent heat of fusion) for this is supplied by the magma itself.
- Since, most magmas are not superheated, this happens by crystallization of primary minerals.
- These minerals would crystallize in the normal course of events, even if there were no assimilation; assimilation simply causes them to crystallize earlier.
- Thus, the amount of assimilation that takes place is normally offset by an almost equivalent amount of crystallization.
- Large quantities of magma containing a significant fraction of assimilated material are therefore not likely to form.
- Assimilation also depends on the way magma rises through the crust.
- If it happens through dikes, the assimilation is minimum. If magma rises by stoping or melting, assimilation of roof rocks may be extensive. Xenoliths are assimilated in this way.
- At first the magma might be entirely mantle-derived, but as it ascends it would steadily be contaminated by crustal material. This process is called zone melting.
- The distance magma can rise by assimilating and crystallizing material is limited by the amount of heat lost to the surroundings.
- The amount of crustal contamination in a mantle-derived magma can be gauged from the degree to which the mantle values are disturbed.
- For this we use isotopic ratios of 87Sr/86Sr and 143Nd/144
- 87Sr/86Sr ratio in crustal rocks is higher than that in mantle-derived rocks.
- 143Nd/144Nd ratio in crustal rocks is lower than in mantle ones.
- Since, isotopic ratios give misleading impressions sometimes, they should be used very carefully.
Effects of assimilation
- The assimilation effects the crystallization of magma.
- It also effects the composition of magma.
- Consolidation, formation, migration and evolution of magma got affected.
- It promotes differentiation in magma.